| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50168052 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_305759 (CHEMBL827097) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Ranise, A; Spallarossa, A; Cesarini, S; Bondavalli, F; Schenone, S; Bruno, O; Menozzi, G; Fossa, P; Mosti, L; La Colla, M; Sanna, G; Murreddu, M; Collu, G; Busonera, B; Marongiu, ME; Pani, A; La Colla, P; Loddo, R Structure-based design, parallel synthesis, structure-activity relationship, and molecular modeling studies of thiocarbamates, new potent non-nucleoside HIV-1 reverse transcriptase inhibitor isosteres of phenethylthiazolylthiourea derivatives. J Med Chem48:3858-73 (2005) [PubMed] Article Ranise, A; Spallarossa, A; Cesarini, S; Bondavalli, F; Schenone, S; Bruno, O; Menozzi, G; Fossa, P; Mosti, L; La Colla, M; Sanna, G; Murreddu, M; Collu, G; Busonera, B; Marongiu, ME; Pani, A; La Colla, P; Loddo, R Structure-based design, parallel synthesis, structure-activity relationship, and molecular modeling studies of thiocarbamates, new potent non-nucleoside HIV-1 reverse transcriptase inhibitor isosteres of phenethylthiazolylthiourea derivatives. J Med Chem48:3858-73 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50168052 |
|---|
| n/a |
|---|
| Name | BDBM50168052 |
|---|
| Synonyms: | (4-Iodo-phenyl)-thiocarbamic acid 2-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-ethyl ester | CHEMBL191910 | O-2-(1,3-dioxoisoindolin-2-yl)ethyl4-iodophenylcarbamothioate | O-[2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)ethyl] (4-iodophenyl)thiocarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H13IN2O3S |
|---|
| Mol. Mass. | 452.266 |
|---|
| SMILES | Ic1ccc(NC(=S)OCCN2C(=O)c3ccccc3C2=O)cc1 |
|---|
| Structure | 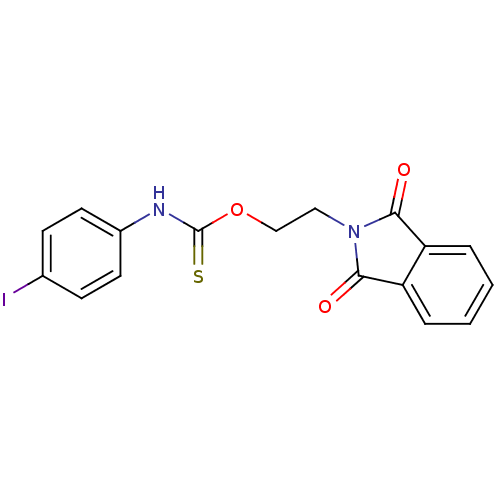
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ranise, A; Spallarossa, A; Cesarini, S; Bondavalli, F; Schenone, S; Bruno, O; Menozzi, G; Fossa, P; Mosti, L; La Colla, M; Sanna, G; Murreddu, M; Collu, G; Busonera, B; Marongiu, ME; Pani, A; La Colla, P; Loddo, R Structure-based design, parallel synthesis, structure-activity relationship, and molecular modeling studies of thiocarbamates, new potent non-nucleoside HIV-1 reverse transcriptase inhibitor isosteres of phenethylthiazolylthiourea derivatives. J Med Chem48:3858-73 (2005) [PubMed] Article
Ranise, A; Spallarossa, A; Cesarini, S; Bondavalli, F; Schenone, S; Bruno, O; Menozzi, G; Fossa, P; Mosti, L; La Colla, M; Sanna, G; Murreddu, M; Collu, G; Busonera, B; Marongiu, ME; Pani, A; La Colla, P; Loddo, R Structure-based design, parallel synthesis, structure-activity relationship, and molecular modeling studies of thiocarbamates, new potent non-nucleoside HIV-1 reverse transcriptase inhibitor isosteres of phenethylthiazolylthiourea derivatives. J Med Chem48:3858-73 (2005) [PubMed] Article