| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 2 |
|---|
| Ligand | BDBM50163114 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303175 (CHEMBL829669) |
|---|
| Ki | 5444±n/a nM |
|---|
| Citation |  Lange, JH; van Stuivenberg, HH; Coolen, HK; Adolfs, TJ; McCreary, AC; Keizer, HG; Wals, HC; Veerman, W; Borst, AJ; de Looff, W; Verveer, PC; Kruse, CG Bioisosteric replacements of the pyrazole moiety of rimonabant: synthesis, biological properties, and molecular modeling investigations of thiazoles, triazoles, and imidazoles as potent and selective CB1 cannabinoid receptor antagonists. J Med Chem48:1823-38 (2005) [PubMed] Article Lange, JH; van Stuivenberg, HH; Coolen, HK; Adolfs, TJ; McCreary, AC; Keizer, HG; Wals, HC; Veerman, W; Borst, AJ; de Looff, W; Verveer, PC; Kruse, CG Bioisosteric replacements of the pyrazole moiety of rimonabant: synthesis, biological properties, and molecular modeling investigations of thiazoles, triazoles, and imidazoles as potent and selective CB1 cannabinoid receptor antagonists. J Med Chem48:1823-38 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 2 |
|---|
| Name: | Cannabinoid receptor 2 |
|---|
| Synonyms: | CANNABINOID CB2 | CB-2 | CB2 | CB2A | CB2B | CNR2 | CNR2_HUMAN | CX5 | Cannabinoid CB2 receptor | Cannabinoid receptor 2 (CB2) | Cannabinoid receptor 2 (CB2R) | hCB2 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 39690.94 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P34972 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MEECWVTEIANGSKDGLDSNPMKDYMILSGPQKTAVAVLCTLLGLLSALENVAVLYLILS
SHQLRRKPSYLFIGSLAGADFLASVVFACSFVNFHVFHGVDSKAVFLLKIGSVTMTFTAS
VGSLLLTAIDRYLCLRYPPSYKALLTRGRALVTLGIMWVLSALVSYLPLMGWTCCPRPCS
ELFPLIPNDYLLSWLLFIAFLFSGIIYTYGHVLWKAHQHVASLSGHQDRQVPGMARMRLD
VRLAKTLGLVLAVLLICWFPVLALMAHSLATTLSDQVKKAFAFCSMLCLINSMVNPVIYA
LRSGEIRSSAHHCLAHWKKCVRGLGSEAKEEAPRSSVTETEADGKITPWPDSRDLDLSDC
|
|
|
|---|
| BDBM50163114 |
|---|
| n/a |
|---|
| Name | BDBM50163114 |
|---|
| Synonyms: | 5-(4-Chloro-phenyl)-1-(2,4-dichloro-phenyl)-1H-[1,2,4]triazole-3-carboxylic acid piperidin-1-ylamide; dihydrochloride | CHEMBL558564 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H18Cl3N5O |
|---|
| Mol. Mass. | 450.749 |
|---|
| SMILES | Clc1ccc(cc1)-c1nc(nn1-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 |
|---|
| Structure | 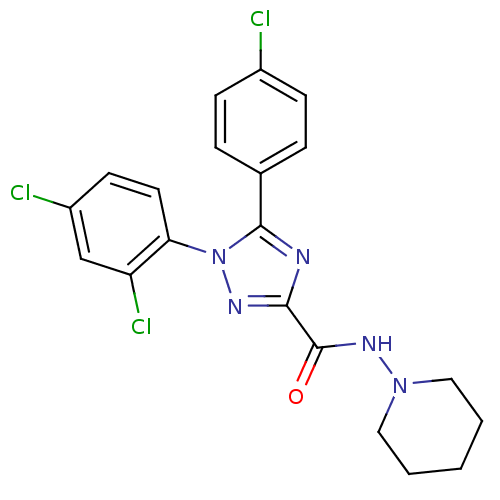
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lange, JH; van Stuivenberg, HH; Coolen, HK; Adolfs, TJ; McCreary, AC; Keizer, HG; Wals, HC; Veerman, W; Borst, AJ; de Looff, W; Verveer, PC; Kruse, CG Bioisosteric replacements of the pyrazole moiety of rimonabant: synthesis, biological properties, and molecular modeling investigations of thiazoles, triazoles, and imidazoles as potent and selective CB1 cannabinoid receptor antagonists. J Med Chem48:1823-38 (2005) [PubMed] Article
Lange, JH; van Stuivenberg, HH; Coolen, HK; Adolfs, TJ; McCreary, AC; Keizer, HG; Wals, HC; Veerman, W; Borst, AJ; de Looff, W; Verveer, PC; Kruse, CG Bioisosteric replacements of the pyrazole moiety of rimonabant: synthesis, biological properties, and molecular modeling investigations of thiazoles, triazoles, and imidazoles as potent and selective CB1 cannabinoid receptor antagonists. J Med Chem48:1823-38 (2005) [PubMed] Article