| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM21281 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_330571 (CHEMBL871146) |
|---|
| Ki | 3802±n/a nM |
|---|
| Citation |  Muccioli, GG; Wouters, J; Charlier, C; Scriba, GK; Pizza, T; Di Pace, P; De Martino, P; Poppitz, W; Poupaert, JH; Lambert, DM Synthesis and activity of 1,3,5-triphenylimidazolidine-2,4-diones and 1,3,5-triphenyl-2-thioxoimidazolidin-4-ones: characterization of new CB1 cannabinoid receptor inverse agonists/antagonists. J Med Chem49:872-82 (2006) [PubMed] Article Muccioli, GG; Wouters, J; Charlier, C; Scriba, GK; Pizza, T; Di Pace, P; De Martino, P; Poppitz, W; Poupaert, JH; Lambert, DM Synthesis and activity of 1,3,5-triphenylimidazolidine-2,4-diones and 1,3,5-triphenyl-2-thioxoimidazolidin-4-ones: characterization of new CB1 cannabinoid receptor inverse agonists/antagonists. J Med Chem49:872-82 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM21281 |
|---|
| n/a |
|---|
| Name | BDBM21281 |
|---|
| Synonyms: | (11R)-2-methyl-11-(morpholin-4-ylmethyl)-3-(naphthalen-1-ylcarbonyl)-9-oxa-1-azatricyclo[6.3.1.0^{4,12}]dodeca-2,4(12),5,7-tetraene | (2,3-dihydro-5-methyl-3-((4-morpholinyl)methyl)pyrrolo-(1,2,3-de)-1,4-benzoxazin-6-yl)(1-naphthalenyl)methanone monomethanesulfonat | CHEMBL188 | WIN 55,212-2 | WIN 55212-2 | WIN-55212 | WIN55212-2 |
|---|
| Type | Analgesic |
|---|
| Emp. Form. | C27H26N2O3 |
|---|
| Mol. Mass. | 426.5069 |
|---|
| SMILES | Cc1c(C(=O)c2cccc3ccccc23)c2cccc3OC[C@@H](CN4CCOCC4)n1c23 |r| |
|---|
| Structure | 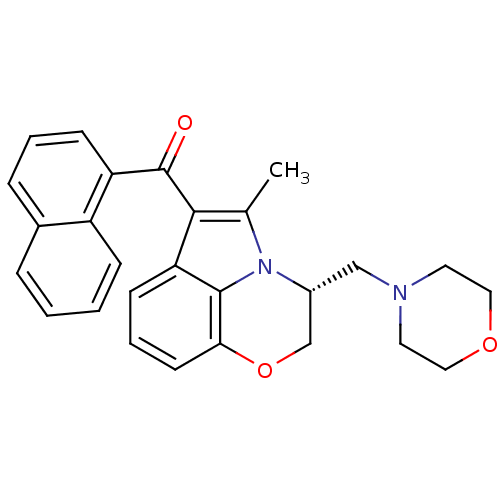
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Muccioli, GG; Wouters, J; Charlier, C; Scriba, GK; Pizza, T; Di Pace, P; De Martino, P; Poppitz, W; Poupaert, JH; Lambert, DM Synthesis and activity of 1,3,5-triphenylimidazolidine-2,4-diones and 1,3,5-triphenyl-2-thioxoimidazolidin-4-ones: characterization of new CB1 cannabinoid receptor inverse agonists/antagonists. J Med Chem49:872-82 (2006) [PubMed] Article
Muccioli, GG; Wouters, J; Charlier, C; Scriba, GK; Pizza, T; Di Pace, P; De Martino, P; Poppitz, W; Poupaert, JH; Lambert, DM Synthesis and activity of 1,3,5-triphenylimidazolidine-2,4-diones and 1,3,5-triphenyl-2-thioxoimidazolidin-4-ones: characterization of new CB1 cannabinoid receptor inverse agonists/antagonists. J Med Chem49:872-82 (2006) [PubMed] Article