| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-X-C chemokine receptor type 3 |
|---|
| Ligand | BDBM50211114 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_438654 (CHEMBL888985) |
|---|
| IC50 | 8±n/a nM |
|---|
| Citation |  Johnson, M; Li, AR; Liu, J; Fu, Z; Zhu, L; Miao, S; Wang, X; Xu, Q; Huang, A; Marcus, A; Xu, F; Ebsworth, K; Sablan, E; Danao, J; Kumer, J; Dairaghi, D; Lawrence, C; Sullivan, T; Tonn, G; Schall, T; Collins, T; Medina, J Discovery and optimization of a series of quinazolinone-derived antagonists of CXCR3. Bioorg Med Chem Lett17:3339-43 (2007) [PubMed] Article Johnson, M; Li, AR; Liu, J; Fu, Z; Zhu, L; Miao, S; Wang, X; Xu, Q; Huang, A; Marcus, A; Xu, F; Ebsworth, K; Sablan, E; Danao, J; Kumer, J; Dairaghi, D; Lawrence, C; Sullivan, T; Tonn, G; Schall, T; Collins, T; Medina, J Discovery and optimization of a series of quinazolinone-derived antagonists of CXCR3. Bioorg Med Chem Lett17:3339-43 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-X-C chemokine receptor type 3 |
|---|
| Name: | C-X-C chemokine receptor type 3 |
|---|
| Synonyms: | AAO92295.1 | C-X-C chemokine receptor type 3 | C-X-C chemokine receptor type 3 (CXCR3) | C-X-C chemokine receptor type 3 (CXCR3A) | CXCR3 | CXCR3A | CXCR3_HUMAN | GPR9 | chemokine (C-X-C motif) receptor 3 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 40665.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 368 |
|---|
| Sequence: | MVLEVSDHQVLNDAEVAALLENFSSSYDYGENESDSCCTSPPCPQDFSLNFDRAFLPALY
SLLFLLGLLGNGAVAAVLLSRRTALSSTDTFLLHLAVADTLLVLTLPLWAVDAAVQWVFG
SGLCKVAGALFNINFYAGALLLACISFDRYLNIVHATQLYRRGPPARVTLTCLAVWGLCL
LFALPDFIFLSAHHDERLNATHCQYNFPQVGRTALRVLQLVAGFLLPLLVMAYCYAHILA
VLLVSRGQRRLRAMRLVVVVVVAFALCWTPYHLVVLVDILMDLGALARNCGRESRVDVAK
SVTSGLGYMHCCLNPLLYAFVGVKFRERMWMLLLRLGCPNQRGLQRQPSSSRRDSSWSET
SEASYSGL
|
|
|
|---|
| BDBM50211114 |
|---|
| n/a |
|---|
| Name | BDBM50211114 |
|---|
| Synonyms: | (-)-(R)-N-(1-(3-(4-ethoxyphenyl)-4-oxo-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl)ethyl)-N-(pyridin-3-ylmethyl)-2-(4-(trifluoromethoxy)phenyl)acetamide | (R)-N-(1-(3-(4-ethoxyphenyl)-4-oxo-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl)ethyl)-N-(pyridin-3-ylmethyl)-2-(4-(trifluoromethoxy)phenyl)acetamide | AMG-487 | CHEMBL397983 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H28F3N5O4 |
|---|
| Mol. Mass. | 603.591 |
|---|
| SMILES | CCOc1ccc(cc1)-n1c(nc2ncccc2c1=O)[C@@H](C)N(Cc1cccnc1)C(=O)Cc1ccc(OC(F)(F)F)cc1 |r| |
|---|
| Structure | 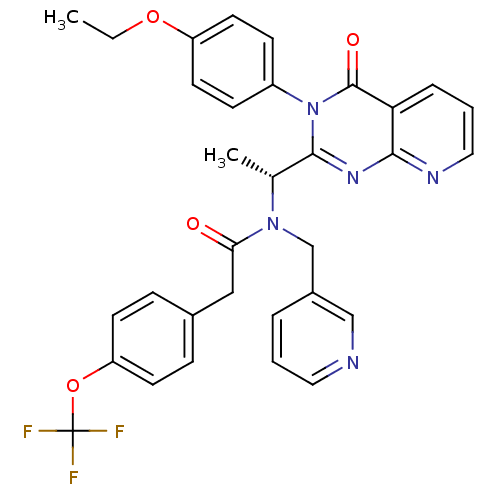
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Johnson, M; Li, AR; Liu, J; Fu, Z; Zhu, L; Miao, S; Wang, X; Xu, Q; Huang, A; Marcus, A; Xu, F; Ebsworth, K; Sablan, E; Danao, J; Kumer, J; Dairaghi, D; Lawrence, C; Sullivan, T; Tonn, G; Schall, T; Collins, T; Medina, J Discovery and optimization of a series of quinazolinone-derived antagonists of CXCR3. Bioorg Med Chem Lett17:3339-43 (2007) [PubMed] Article
Johnson, M; Li, AR; Liu, J; Fu, Z; Zhu, L; Miao, S; Wang, X; Xu, Q; Huang, A; Marcus, A; Xu, F; Ebsworth, K; Sablan, E; Danao, J; Kumer, J; Dairaghi, D; Lawrence, C; Sullivan, T; Tonn, G; Schall, T; Collins, T; Medina, J Discovery and optimization of a series of quinazolinone-derived antagonists of CXCR3. Bioorg Med Chem Lett17:3339-43 (2007) [PubMed] Article