| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50194264 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_458384 (CHEMBL942703) |
|---|
| IC50 | 150±n/a nM |
|---|
| Citation |  VanAlstine, MA; Wentland, MP; Cohen, DJ; Bidlack, JM Redefining the structure-activity relationships of 2,6-methano-3-benzazocines. 5. Opioid receptor binding properties of N-((4'-phenyl)-phenethyl) analogues of 8-CAC. Bioorg Med Chem Lett17:6516-20 (2007) [PubMed] Article VanAlstine, MA; Wentland, MP; Cohen, DJ; Bidlack, JM Redefining the structure-activity relationships of 2,6-methano-3-benzazocines. 5. Opioid receptor binding properties of N-((4'-phenyl)-phenethyl) analogues of 8-CAC. Bioorg Med Chem Lett17:6516-20 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | M-OR-1 | MOP | MOR-1 | MOR1 | MUOR1 | Mu Opioid Receptor | Mu opiate receptor | OPIATE Mu | OPRM1 | OPRM_HUMAN | hMOP | mu-type opioid receptor isoform MOR-1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44789.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35372 |
|---|
| Residue: | 400 |
|---|
| Sequence: | MDSSAAPTNASNCTDALAYSSCSPAPSPGSWVNLSHLDGNLSDPCGPNRTDLGGRDSLCP
PTGSPSMITAITIMALYSIVCVVGLFGNFLVMYVIVRYTKMKTATNIYIFNLALADALAT
STLPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSIFTLCTMSVDRYIAVCHPVKALDF
RTPRNAKIINVCNWILSSAIGLPVMFMATTKYRQGSIDCTLTFSHPTWYWENLLKICVFI
FAFIMPVLIITVCYGLMILRLKSVRMLSGSKEKDRNLRRITRMVLVVVAVFIVCWTPIHI
YVIIKALVTIPETTFQTVSWHFCIALGYTNSCLNPVLYAFLDENFKRCFREFCIPTSSNI
EQQNSTRIRQNTRDHPSTANTVDRTNHQLENLEAETAPLP
|
|
|
|---|
| BDBM50194264 |
|---|
| n/a |
|---|
| Name | BDBM50194264 |
|---|
| Synonyms: | (+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-cis-6,11-dimethyl-N-(2-[1,1'-biphenyl]-4-ylethyl)-2,6-methano-3-benzazocine-8-carboxamide | (6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4,5,6-hexahydro-2,6-methano-benzo[d]azocine-8-carboxylic acid (2-biphenyl-4-yl-ethyl)-amide | CHEMBL377789 | US9656961, Example 00154 | cis-rac-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4,5,6-hexahydro-2,6-methano-benzo[d]azocine-8-carboxylic acid (2-biphenyl-4-yl-ethyl)-amide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H38N2O |
|---|
| Mol. Mass. | 478.6676 |
|---|
| SMILES | C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| |
|---|
| Structure | 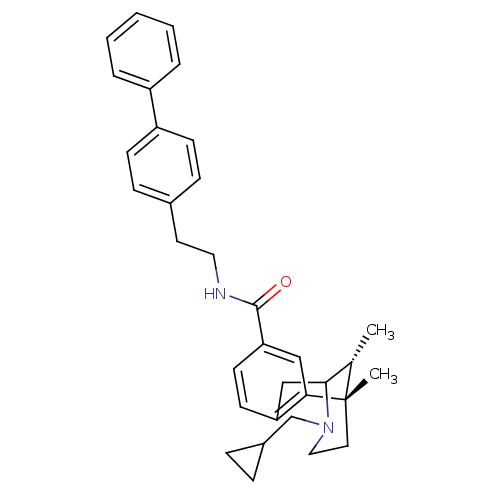
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 VanAlstine, MA; Wentland, MP; Cohen, DJ; Bidlack, JM Redefining the structure-activity relationships of 2,6-methano-3-benzazocines. 5. Opioid receptor binding properties of N-((4'-phenyl)-phenethyl) analogues of 8-CAC. Bioorg Med Chem Lett17:6516-20 (2007) [PubMed] Article
VanAlstine, MA; Wentland, MP; Cohen, DJ; Bidlack, JM Redefining the structure-activity relationships of 2,6-methano-3-benzazocines. 5. Opioid receptor binding properties of N-((4'-phenyl)-phenethyl) analogues of 8-CAC. Bioorg Med Chem Lett17:6516-20 (2007) [PubMed] Article