| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin receptor type A |
|---|
| Ligand | BDBM50212239 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_460796 (CHEMBL944737) |
|---|
| IC50 | >2000±n/a nM |
|---|
| Citation |  Kort, ME; Drizin, I; Gregg, RJ; Scanio, MJ; Shi, L; Gross, MF; Atkinson, RN; Johnson, MS; Pacofsky, GJ; Thomas, JB; Carroll, WA; Krambis, MJ; Liu, D; Shieh, CC; Zhang, X; Hernandez, G; Mikusa, JP; Zhong, C; Joshi, S; Honore, P; Roeloffs, R; Marsh, KC; Murray, BP; Liu, J; Werness, S; Faltynek, CR; Krafte, DS; Jarvis, MF; Chapman, ML; Marron, BE Discovery and biological evaluation of 5-aryl-2-furfuramides, potent and selective blockers of the Nav1.8 sodium channel with efficacy in models of neuropathic and inflammatory pain. J Med Chem51:407-16 (2008) [PubMed] Article Kort, ME; Drizin, I; Gregg, RJ; Scanio, MJ; Shi, L; Gross, MF; Atkinson, RN; Johnson, MS; Pacofsky, GJ; Thomas, JB; Carroll, WA; Krambis, MJ; Liu, D; Shieh, CC; Zhang, X; Hernandez, G; Mikusa, JP; Zhong, C; Joshi, S; Honore, P; Roeloffs, R; Marsh, KC; Murray, BP; Liu, J; Werness, S; Faltynek, CR; Krafte, DS; Jarvis, MF; Chapman, ML; Marron, BE Discovery and biological evaluation of 5-aryl-2-furfuramides, potent and selective blockers of the Nav1.8 sodium channel with efficacy in models of neuropathic and inflammatory pain. J Med Chem51:407-16 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholecystokinin receptor type A |
|---|
| Name: | Cholecystokinin receptor type A |
|---|
| Synonyms: | CCK-A receptor | CCK-AR | CCK1-R | CCKAR | CCKAR_HUMAN | CCKRA | Cholecystokinin receptor | Cholecystokinin receptor type A | Cholecystokinin-1 Receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 47859.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Stable expression of human CCK-1 receptors in HEK 293 cells. |
|---|
| Residue: | 428 |
|---|
| Sequence: | MDVVDSLLVNGSNITPPCELGLENETLFCLDQPRPSKEWQPAVQILLYSLIFLLSVLGNT
LVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLKDFIFGSAVCKTTTYF
MGTSVSVSTFNLVAISLERYGAICKPLQSRVWQTKSHALKVIAATWCLSFTIMTPYPIYS
NLVPFTKNNNQTANMCRFLLPNDVMQQSWHTFLLLILFLIPGIVMMVAYGLISLELYQGI
KFEASQKKSAKERKPSTTSSGKYEDSDGCYLQKTRPPRKLELRQLSTGSSSRANRIRSNS
SAANLMAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTASAERRLSGTPISFILLLSY
TSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGARGEVGEEEEGGTTGASLSRFSYSH
MSASVPPQ
|
|
|
|---|
| BDBM50212239 |
|---|
| n/a |
|---|
| Name | BDBM50212239 |
|---|
| Synonyms: | 5-(4-chlorophenyl)-N-(3,5-dimethoxyphenyl)furan-2-carboxamide | A-803467 | CHEMBL250699 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H16ClNO4 |
|---|
| Mol. Mass. | 357.788 |
|---|
| SMILES | COc1cc(NC(=O)c2ccc(o2)-c2ccc(Cl)cc2)cc(OC)c1 |
|---|
| Structure | 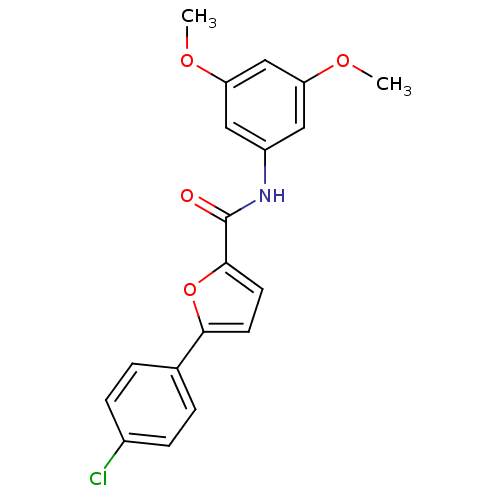
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kort, ME; Drizin, I; Gregg, RJ; Scanio, MJ; Shi, L; Gross, MF; Atkinson, RN; Johnson, MS; Pacofsky, GJ; Thomas, JB; Carroll, WA; Krambis, MJ; Liu, D; Shieh, CC; Zhang, X; Hernandez, G; Mikusa, JP; Zhong, C; Joshi, S; Honore, P; Roeloffs, R; Marsh, KC; Murray, BP; Liu, J; Werness, S; Faltynek, CR; Krafte, DS; Jarvis, MF; Chapman, ML; Marron, BE Discovery and biological evaluation of 5-aryl-2-furfuramides, potent and selective blockers of the Nav1.8 sodium channel with efficacy in models of neuropathic and inflammatory pain. J Med Chem51:407-16 (2008) [PubMed] Article
Kort, ME; Drizin, I; Gregg, RJ; Scanio, MJ; Shi, L; Gross, MF; Atkinson, RN; Johnson, MS; Pacofsky, GJ; Thomas, JB; Carroll, WA; Krambis, MJ; Liu, D; Shieh, CC; Zhang, X; Hernandez, G; Mikusa, JP; Zhong, C; Joshi, S; Honore, P; Roeloffs, R; Marsh, KC; Murray, BP; Liu, J; Werness, S; Faltynek, CR; Krafte, DS; Jarvis, MF; Chapman, ML; Marron, BE Discovery and biological evaluation of 5-aryl-2-furfuramides, potent and selective blockers of the Nav1.8 sodium channel with efficacy in models of neuropathic and inflammatory pain. J Med Chem51:407-16 (2008) [PubMed] Article