| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lanosterol 14-alpha demethylase |
|---|
| Ligand | BDBM35346 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_470779 (CHEMBL934981) |
|---|
| IC50 | 4000±n/a nM |
|---|
| Citation |  Ekins, S; Mankowski, DC; Hoover, DJ; Lawton, MP; Treadway, JL; Harwood, HJ Three-dimensional quantitative structure-activity relationship analysis of human CYP51 inhibitors. Drug Metab Dispos35:493-500 (2007) [PubMed] Article Ekins, S; Mankowski, DC; Hoover, DJ; Lawton, MP; Treadway, JL; Harwood, HJ Three-dimensional quantitative structure-activity relationship analysis of human CYP51 inhibitors. Drug Metab Dispos35:493-500 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lanosterol 14-alpha demethylase |
|---|
| Name: | Lanosterol 14-alpha demethylase |
|---|
| Synonyms: | CP51A_HUMAN | CYP51 | CYP51A1 | CYPLI | Cytochrome P450 51 | Cytochrome P450 51A1 | LDM | Lanosterol 14-alpha demethylase | P450-14DM | P45014DM | P450LI | Sterol 14α-demethylase (CYP51) | Sterol 14-alpha demethylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 56817.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16850 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MLLLGLLQAGGSVLGQAMEKVTGGNLLSMLLIACAFTLSLVYLIRLAAGHLVQLPAGVKS
PPYIFSPIPFLGHAIAFGKSPIEFLENAYEKYGPVFSFTMVGKTFTYLLGSDAAALLFNS
KNEDLNAEDVYSRLTTPVFGKGVAYDVPNPVFLEQKKMLKSGLNIAHFKQHVSIIEKETK
EYFESWGESGEKNVFEALSELIILTASHCLHGKEIRSQLNEKVAQLYADLDGGFSHAAWL
LPGWLPLPSFRRRDRAHREIKDIFYKAIQKRRQSQEKIDDILQTLLDATYKDGRPLTDDE
VAGMLIGLLLAGQHTSSTTSAWMGFFLARDKTLQKKCYLEQKTVCGENLPPLTYDQLKDL
NLLDRCIKETLRLRPPIMIMMRMARTPQTVAGYTIPPGHQVCVSPTVNQRLKDSWVERLD
FNPDRYLQDNPASGEKFAYVPFGAGRHRCIGENFAYVQIKTIWSTMLRLYEFDLIDGYFP
TVNYTTMIHTPENPVIRYKRRSK
|
|
|
|---|
| BDBM35346 |
|---|
| n/a |
|---|
| Name | BDBM35346 |
|---|
| Synonyms: | (S)-5-chloro-N-(3-(4-fluorophenyl)-1-(4-hydroxypiperidin-1-yl)-1-oxopropan-2-yl)-1H-indole-2-carboxamide | 5-CHLORO-1H-INDOLE-2-CARBOXYLIC ACID [1-(4-FLUOROBENZYL)-2-(4-HYDROXYPIPERIDIN-1YL)-2-OXOETHYL]AMIDE | 5-Chloro-1H-indole-2-carboxylic acid [(S)-1-(4-fluoro-benzyl)-2-(4-hydroxy-piperidin-1-yl)-2-oxo-ethyl]-amide | CHEMBL99889 | CP-320626 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23ClFN3O3 |
|---|
| Mol. Mass. | 443.898 |
|---|
| SMILES | OC1CCN(CC1)C(=O)[C@H](Cc1ccc(F)cc1)NC(=O)c1cc2cc(Cl)ccc2[nH]1 |r| |
|---|
| Structure | 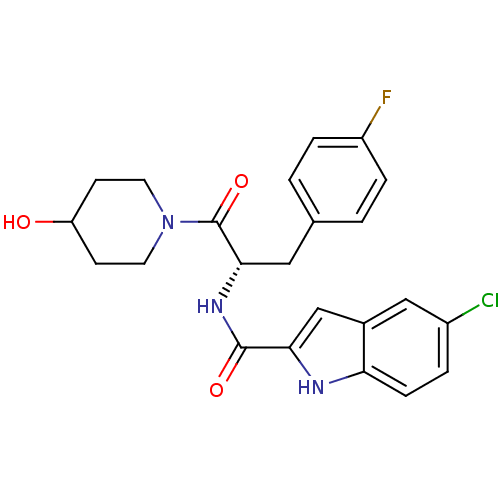
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ekins, S; Mankowski, DC; Hoover, DJ; Lawton, MP; Treadway, JL; Harwood, HJ Three-dimensional quantitative structure-activity relationship analysis of human CYP51 inhibitors. Drug Metab Dispos35:493-500 (2007) [PubMed] Article
Ekins, S; Mankowski, DC; Hoover, DJ; Lawton, MP; Treadway, JL; Harwood, HJ Three-dimensional quantitative structure-activity relationship analysis of human CYP51 inhibitors. Drug Metab Dispos35:493-500 (2007) [PubMed] Article