| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen synthase kinase-3 beta |
|---|
| Ligand | BDBM7478 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_563806 (CHEMBL994318) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Gray, NS; Wodicka, L; Thunnissen, AM; Norman, TC; Kwon, S; Espinoza, FH; Morgan, DO; Barnes, G; LeClerc, S; Meijer, L; Kim, SH; Lockhart, DJ; Schultz, PG Exploiting chemical libraries, structure, and genomics in the search for kinase inhibitors. Science281:533-538 (1998) [PubMed] Gray, NS; Wodicka, L; Thunnissen, AM; Norman, TC; Kwon, S; Espinoza, FH; Morgan, DO; Barnes, G; LeClerc, S; Meijer, L; Kim, SH; Lockhart, DJ; Schultz, PG Exploiting chemical libraries, structure, and genomics in the search for kinase inhibitors. Science281:533-538 (1998) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen synthase kinase-3 beta |
|---|
| Name: | Glycogen synthase kinase-3 beta |
|---|
| Synonyms: | GSK-3 beta | GSK-3, beta | GSK3B | GSK3B_HUMAN | Glycogen synthase kinase 3 beta (GSK3B) | Glycogen synthase kinase 3-beta (GSK3B) | Glycogen synthase kinase-3 beta (GSK-3B) | Glycogen synthase kinase-3 beta (GSK3 Beta) | Glycogen synthase kinase-3 beta (GSK3B) | Glycogen synthase kinase-3B (GSK-3B) | Glycogen synthase kinase-3beta (GSK3B) | Serine/threonine-protein kinase GSK3B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 46756.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49841 |
|---|
| Residue: | 420 |
|---|
| Sequence: | MSGRPRTTSFAESCKPVQQPSAFGSMKVSRDKDGSKVTTVVATPGQGPDRPQEVSYTDTK
VIGNGSFGVVYQAKLCDSGELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFYSSG
EKKDEVYLNLVLDYVPETVYRVARHYSRAKQTLPVIYVKLYMYQLFRSLAYIHSFGICHR
DIKPQNLLLDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSSIDV
WSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIKAHP
WTKVFRPRTPPEAIALCSRLLEYTPTARLTPLEACAHSFFDELRDPNVKLPNGRDTPALF
NFTTQELSSNPPLATILIPPHARIQAAASTPTNATAASDANTGDRGQTNNAASASASNST
|
|
|
|---|
| BDBM7478 |
|---|
| n/a |
|---|
| Name | BDBM7478 |
|---|
| Synonyms: | 2-chloro-4-[(2-{[(1R)-1-(hydroxymethyl)-2-methylpropyl]amino}-9-isopropyl-9H-purin-6-yl)amino]benzoic acid | 2-chloro-4-[(2-{[(2R)-1-hydroxy-3-methylbutan-2-yl]amino}-9-(propan-2-yl)-9H-purin-6-yl)amino]benzoic acid | CHEMBL23254 | Purvalanol B |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H25ClN6O3 |
|---|
| Mol. Mass. | 432.904 |
|---|
| SMILES | CC(C)[C@H](CO)Nc1nc(Nc2ccc(C(O)=O)c(Cl)c2)c2ncn(C(C)C)c2n1 |r| |
|---|
| Structure | 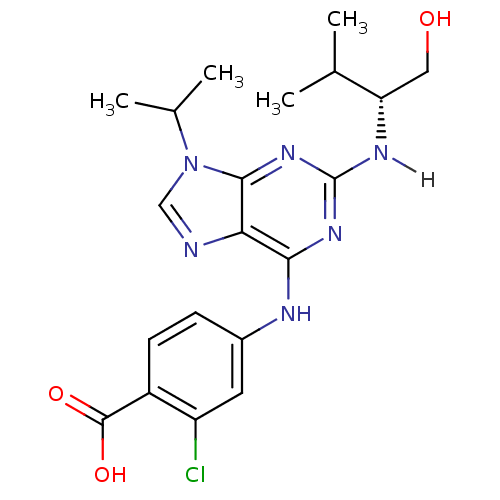
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gray, NS; Wodicka, L; Thunnissen, AM; Norman, TC; Kwon, S; Espinoza, FH; Morgan, DO; Barnes, G; LeClerc, S; Meijer, L; Kim, SH; Lockhart, DJ; Schultz, PG Exploiting chemical libraries, structure, and genomics in the search for kinase inhibitors. Science281:533-538 (1998) [PubMed]
Gray, NS; Wodicka, L; Thunnissen, AM; Norman, TC; Kwon, S; Espinoza, FH; Morgan, DO; Barnes, G; LeClerc, S; Meijer, L; Kim, SH; Lockhart, DJ; Schultz, PG Exploiting chemical libraries, structure, and genomics in the search for kinase inhibitors. Science281:533-538 (1998) [PubMed]