| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50319822 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_634880 (CHEMBL1120512) |
|---|
| IC50 | 17700±n/a nM |
|---|
| Citation |  Fray, MJ; Fish, PV; Allan, GA; Bish, G; Clarke, N; Eccles, R; Harrison, AC; Le Net, JL; Phillips, SC; Regan, N; Sobry, C; Stobie, A; Wakenhut, F; Westbrook, D; Westbrook, SL; Whitlock, GA Second generation N-(1,2-diphenylethyl)piperazines as dual serotonin and noradrenaline reuptake inhibitors: improving metabolic stability and reducing ion channel activity. Bioorg Med Chem Lett20:3788-92 (2010) [PubMed] Article Fray, MJ; Fish, PV; Allan, GA; Bish, G; Clarke, N; Eccles, R; Harrison, AC; Le Net, JL; Phillips, SC; Regan, N; Sobry, C; Stobie, A; Wakenhut, F; Westbrook, D; Westbrook, SL; Whitlock, GA Second generation N-(1,2-diphenylethyl)piperazines as dual serotonin and noradrenaline reuptake inhibitors: improving metabolic stability and reducing ion channel activity. Bioorg Med Chem Lett20:3788-92 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50319822 |
|---|
| n/a |
|---|
| Name | BDBM50319822 |
|---|
| Synonyms: | 1-(1-(2-fluorophenyl)-2-(2-methoxyphenyl)ethyl)piperazine | CHEMBL1085959 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H23FN2O |
|---|
| Mol. Mass. | 314.3971 |
|---|
| SMILES | COc1ccccc1CC(N1CCNCC1)c1ccccc1F |
|---|
| Structure | 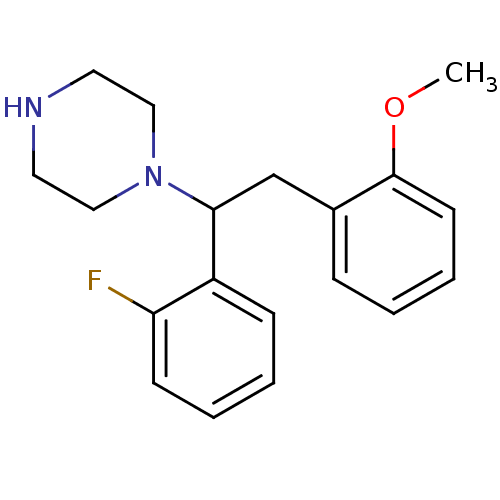
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fray, MJ; Fish, PV; Allan, GA; Bish, G; Clarke, N; Eccles, R; Harrison, AC; Le Net, JL; Phillips, SC; Regan, N; Sobry, C; Stobie, A; Wakenhut, F; Westbrook, D; Westbrook, SL; Whitlock, GA Second generation N-(1,2-diphenylethyl)piperazines as dual serotonin and noradrenaline reuptake inhibitors: improving metabolic stability and reducing ion channel activity. Bioorg Med Chem Lett20:3788-92 (2010) [PubMed] Article
Fray, MJ; Fish, PV; Allan, GA; Bish, G; Clarke, N; Eccles, R; Harrison, AC; Le Net, JL; Phillips, SC; Regan, N; Sobry, C; Stobie, A; Wakenhut, F; Westbrook, D; Westbrook, SL; Whitlock, GA Second generation N-(1,2-diphenylethyl)piperazines as dual serotonin and noradrenaline reuptake inhibitors: improving metabolic stability and reducing ion channel activity. Bioorg Med Chem Lett20:3788-92 (2010) [PubMed] Article