| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H4 receptor |
|---|
| Ligand | BDBM50317865 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_643318 (CHEMBL1177489) |
|---|
| Ki | 1.1±n/a nM |
|---|
| Citation |  Ishikawa, M; Furuuchi, T; Yamauchi, M; Yokoyama, F; Kakui, N; Sato, Y Synthesis and structure-activity relationships of N-aryl-piperidine derivatives as potent (partial) agonists for human histamine H3 receptor. Bioorg Med Chem18:5441-8 (2010) [PubMed] Article Ishikawa, M; Furuuchi, T; Yamauchi, M; Yokoyama, F; Kakui, N; Sato, Y Synthesis and structure-activity relationships of N-aryl-piperidine derivatives as potent (partial) agonists for human histamine H3 receptor. Bioorg Med Chem18:5441-8 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H4 receptor |
|---|
| Name: | Histamine H4 receptor |
|---|
| Synonyms: | AXOR35 | G-protein coupled receptor 105 | GPCR105 | GPRv53 | HH4R | HISTAMINE H4 | HRH4 | HRH4_HUMAN | Histamine H4 receptor | Histamine H4 receptor (H4R) | Histamine receptor (H3 and H4) | Pfi-013 | SP9144 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44517.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH4R receptors. |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPDTNSTINLSLSTRVTLAFFMSLVAFAIMLGNALVILAFVVDKNLRHRSSYFFLNLAIS
DFFVGVISIPLYIPHTLFEWDFGKEICVFWLTTDYLLCTASVYNIVLISYDRYLSVSNAV
SYRTQHTGVLKIVTLMVAVWVLAFLVNGPMILVSESWKDEGSECEPGFFSEWYILAITSF
LEFVIPVILVAYFNMNIYWSLWKRDHLSRCQSHPGLTAVSSNICGHSFRGRLSSRRSLSA
STEVPASFHSERQRRKSSLMFSSRTKMNSNTIASKMGSFSQSDSVALHQREHVELLRARR
LAKSLAILLGVFAVCWAPYSLFTIVLSFYSSATGPKSVWYRIAFWLQWFNSFVNPLLYPL
CHKRFQKAFLKIFCIKKQPLPSQHSRSVSS
|
|
|
|---|
| BDBM50317865 |
|---|
| n/a |
|---|
| Name | BDBM50317865 |
|---|
| Synonyms: | 2-(2-(4-tert-Butylphenylthio)ethyl)-1H-imidazole | 4-(2-(4-tert-butylphenylthio)ethyl)-1H-imidazole | CHEMBL1098230 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H20N2S |
|---|
| Mol. Mass. | 260.398 |
|---|
| SMILES | CC(C)(C)c1ccc(SCCc2cnc[nH]2)cc1 |
|---|
| Structure | 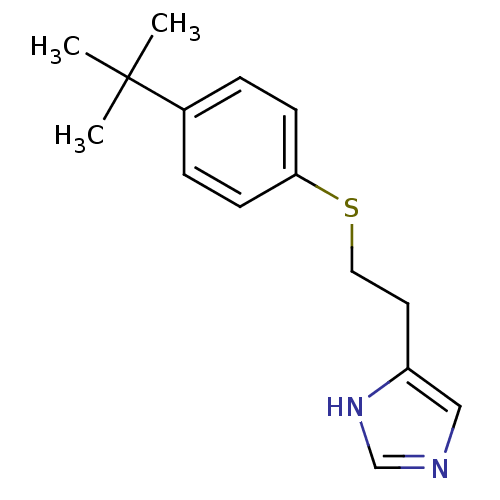
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ishikawa, M; Furuuchi, T; Yamauchi, M; Yokoyama, F; Kakui, N; Sato, Y Synthesis and structure-activity relationships of N-aryl-piperidine derivatives as potent (partial) agonists for human histamine H3 receptor. Bioorg Med Chem18:5441-8 (2010) [PubMed] Article
Ishikawa, M; Furuuchi, T; Yamauchi, M; Yokoyama, F; Kakui, N; Sato, Y Synthesis and structure-activity relationships of N-aryl-piperidine derivatives as potent (partial) agonists for human histamine H3 receptor. Bioorg Med Chem18:5441-8 (2010) [PubMed] Article