| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 14 |
|---|
| Ligand | BDBM50323660 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_645060 (CHEMBL1211566) |
|---|
| IC50 | 25±n/a nM |
|---|
| Citation |  Montalban, AG; Boman, E; Chang, CD; Ceide, SC; Dahl, R; Dalesandro, D; Delaet, NG; Erb, E; Ernst, JT; Gibbs, A; Kahl, J; Kessler, L; Kucharski, J; Lum, C; Lundström, J; Miller, S; Nakanishi, H; Roberts, E; Saiah, E; Sullivan, R; Urban, J; Wang, Z; Larson, CJ Optimization of alpha-ketoamide based p38 inhibitors through modifications to the region that binds to the allosteric site. Bioorg Med Chem Lett20:4819-24 (2010) [PubMed] Article Montalban, AG; Boman, E; Chang, CD; Ceide, SC; Dahl, R; Dalesandro, D; Delaet, NG; Erb, E; Ernst, JT; Gibbs, A; Kahl, J; Kessler, L; Kucharski, J; Lum, C; Lundström, J; Miller, S; Nakanishi, H; Roberts, E; Saiah, E; Sullivan, R; Urban, J; Wang, Z; Larson, CJ Optimization of alpha-ketoamide based p38 inhibitors through modifications to the region that binds to the allosteric site. Bioorg Med Chem Lett20:4819-24 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 14 |
|---|
| Name: | Mitogen-activated protein kinase 14 |
|---|
| Synonyms: | CSAID-binding protein | CSBP | CSBP1 | CSBP2 | CSPB1 | Cytokine suppressive anti-inflammatory drug-binding protein | MAP kinase 14 | MAP kinase MXI2 | MAP kinase p38 alpha | MAPK 14 | MAPK14 | MAX-interacting protein 2 | MK14_HUMAN | MXI2 | Mitogen-activated protein kinase p38 alpha | SAPK2A | Stress-activated protein kinase 2a | p38 MAP kinase alpha/beta |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 41286.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16539 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGLRVAVKKLSRPFQ
SIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQ
KLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVG
TPGAELLKKISSESARNYIQSLTQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAA
QALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
|
|
|
|---|
| BDBM50323660 |
|---|
| n/a |
|---|
| Name | BDBM50323660 |
|---|
| Synonyms: | CHEMBL1208891 | N-(5-tert-butyl-2-methoxy-3-(methylsulfonamido)phenyl)-2-oxo-2-(4-(pyridin-4-ylamino)naphthalen-1-yl)acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H30N4O5S |
|---|
| Mol. Mass. | 546.637 |
|---|
| SMILES | COc1c(NC(=O)C(=O)c2ccc(Nc3ccncc3)c3ccccc23)cc(cc1NS(C)(=O)=O)C(C)(C)C |
|---|
| Structure | 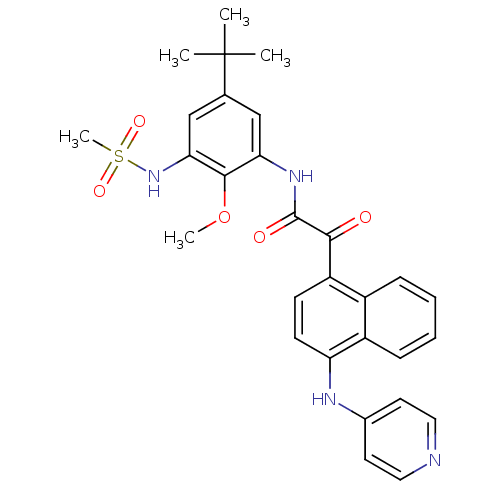
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Montalban, AG; Boman, E; Chang, CD; Ceide, SC; Dahl, R; Dalesandro, D; Delaet, NG; Erb, E; Ernst, JT; Gibbs, A; Kahl, J; Kessler, L; Kucharski, J; Lum, C; Lundström, J; Miller, S; Nakanishi, H; Roberts, E; Saiah, E; Sullivan, R; Urban, J; Wang, Z; Larson, CJ Optimization of alpha-ketoamide based p38 inhibitors through modifications to the region that binds to the allosteric site. Bioorg Med Chem Lett20:4819-24 (2010) [PubMed] Article
Montalban, AG; Boman, E; Chang, CD; Ceide, SC; Dahl, R; Dalesandro, D; Delaet, NG; Erb, E; Ernst, JT; Gibbs, A; Kahl, J; Kessler, L; Kucharski, J; Lum, C; Lundström, J; Miller, S; Nakanishi, H; Roberts, E; Saiah, E; Sullivan, R; Urban, J; Wang, Z; Larson, CJ Optimization of alpha-ketoamide based p38 inhibitors through modifications to the region that binds to the allosteric site. Bioorg Med Chem Lett20:4819-24 (2010) [PubMed] Article