| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calcium/calmodulin-dependent protein kinase type IV |
|---|
| Ligand | BDBM50335188 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_701560 (CHEMBL1656199) |
|---|
| IC50 | >100±n/a nM |
|---|
| Citation |  Kerekes, AD; Esposite, SJ; Tagat, JR; Xiao, Y; Terracina, GA Aurora kinase inhibitors based on the imidazo[1,2-a]pyrazine core: fluorine and deuterium incorporation improve oral absorption and exposure. J Med Chem54:201-10 (2011) [PubMed] Article Kerekes, AD; Esposite, SJ; Tagat, JR; Xiao, Y; Terracina, GA Aurora kinase inhibitors based on the imidazo[1,2-a]pyrazine core: fluorine and deuterium incorporation improve oral absorption and exposure. J Med Chem54:201-10 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calcium/calmodulin-dependent protein kinase type IV |
|---|
| Name: | Calcium/calmodulin-dependent protein kinase type IV |
|---|
| Synonyms: | CAM kinase-GR | CAMK | CAMK-GR | CAMK4 | CAMKIV | CaM kinase IV | CaMK IV | Calcium/calmodulin-dependent protein kinase type IV | KCC4_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 51919.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1459854 |
|---|
| Residue: | 473 |
|---|
| Sequence: | MLKVTVPSCSASSCSSVTASAAPGTASLVPDYWIDGSNRDALSDFFEVESELGRGATSIV
YRCKQKGTQKPYALKVLKKTVDKKIVRTEIGVLLRLSHPNIIKLKEIFETPTEISLVLEL
VTGGELFDRIVEKGYYSERDAADAVKQILEAVAYLHENGIVHRDLKPENLLYATPAPDAP
LKIADFGLSKIVEHQVLMKTVCGTPGYCAPEILRGCAYGPEVDMWSVGIITYILLCGFEP
FYDERGDQFMFRRILNCEYYFISPWWDEVSLNAKDLVRKLIVLDPKKRLTTFQALQHPWV
TGKAANFVHMDTAQKKLQEFNARRKLKAAVKAVVASSRLGSASSSHGSIQESHKASRDPS
PIQDGNEDMKAIPEGEKIQGDGAQAAVKGAQAELMKVQALEKVKGADINAEEAPKMVPKA
VEDGIKVADLELEEGLAEEKLKTVEEAAAPREGQGSSAVGFEVPQQDVILPEY
|
|
|
|---|
| BDBM50335188 |
|---|
| n/a |
|---|
| Name | BDBM50335188 |
|---|
| Synonyms: | CHEMBL1650545 | CHEMBL1650551 | N-[3-[(3,3-Difluoro-1-pyrrolidinyl)methyl]-5-isothiazolyl]-6-methyl-3-(1H-pyrazol-4-yl)imidazo[1,2-a]pyrazin-8-amine Hydrochloride |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H18F2N8S |
|---|
| Mol. Mass. | 416.451 |
|---|
| SMILES | Cc1cn2c(cnc2c(Nc2cc(CN3CCC(F)(F)C3)ns2)n1)-c1cn[nH]c1 |
|---|
| Structure | 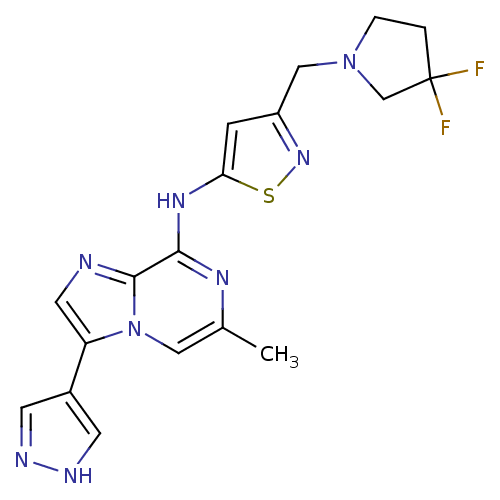
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kerekes, AD; Esposite, SJ; Tagat, JR; Xiao, Y; Terracina, GA Aurora kinase inhibitors based on the imidazo[1,2-a]pyrazine core: fluorine and deuterium incorporation improve oral absorption and exposure. J Med Chem54:201-10 (2011) [PubMed] Article
Kerekes, AD; Esposite, SJ; Tagat, JR; Xiao, Y; Terracina, GA Aurora kinase inhibitors based on the imidazo[1,2-a]pyrazine core: fluorine and deuterium incorporation improve oral absorption and exposure. J Med Chem54:201-10 (2011) [PubMed] Article