| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM50336843 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_718860 (CHEMBL1681242) |
|---|
| Ki | >10000±n/a nM |
|---|
| Citation |  Siméon, FG; Wendahl, MT; Pike, VW Syntheses of 2-amino and 2-halothiazole derivatives as high-affinity metabotropic glutamate receptor subtype 5 ligands and potential radioligands for in vivo imaging. J Med Chem54:901-8 (2012) [PubMed] Article Siméon, FG; Wendahl, MT; Pike, VW Syntheses of 2-amino and 2-halothiazole derivatives as high-affinity metabotropic glutamate receptor subtype 5 ligands and potential radioligands for in vivo imaging. J Med Chem54:901-8 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM50336843 |
|---|
| n/a |
|---|
| Name | BDBM50336843 |
|---|
| Synonyms: | 3-((2-Fluorothiazol-4-yl)ethynyl)-5-fluorobenzonitrile | CHEMBL1672288 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H4F2N2S |
|---|
| Mol. Mass. | 246.235 |
|---|
| SMILES | Fc1nc(cs1)C#Cc1cc(F)cc(c1)C#N |
|---|
| Structure | 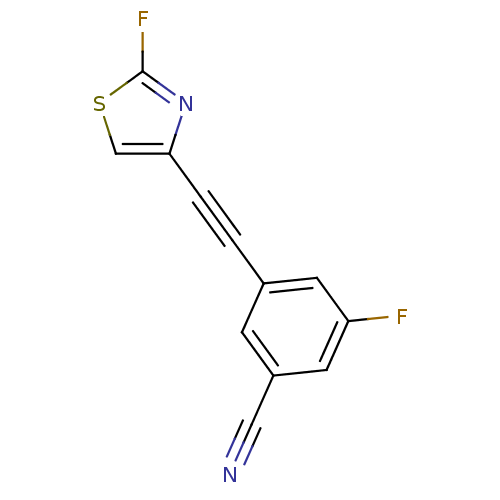
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Siméon, FG; Wendahl, MT; Pike, VW Syntheses of 2-amino and 2-halothiazole derivatives as high-affinity metabotropic glutamate receptor subtype 5 ligands and potential radioligands for in vivo imaging. J Med Chem54:901-8 (2012) [PubMed] Article
Siméon, FG; Wendahl, MT; Pike, VW Syntheses of 2-amino and 2-halothiazole derivatives as high-affinity metabotropic glutamate receptor subtype 5 ligands and potential radioligands for in vivo imaging. J Med Chem54:901-8 (2012) [PubMed] Article