| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50347656 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_756007 (CHEMBL1803983) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Katz, JD; Jewell, JP; Guerin, DJ; Lim, J; Dinsmore, CJ; Deshmukh, SV; Pan, BS; Marshall, CG; Lu, W; Altman, MD; Dahlberg, WK; Davis, L; Falcone, D; Gabarda, AE; Hang, G; Hatch, H; Holmes, R; Kunii, K; Lumb, KJ; Lutterbach, B; Mathvink, R; Nazef, N; Patel, SB; Qu, X; Reilly, JF; Rickert, KW; Rosenstein, C; Soisson, SM; Spencer, KB; Szewczak, AA; Walker, D; Wang, W; Young, J; Zeng, Q Discovery of a 5H-benzo[4,5]cyclohepta[1,2-b]pyridin-5-one (MK-2461) inhibitor of c-Met kinase for the treatment of cancer. J Med Chem54:4092-108 (2011) [PubMed] Article Katz, JD; Jewell, JP; Guerin, DJ; Lim, J; Dinsmore, CJ; Deshmukh, SV; Pan, BS; Marshall, CG; Lu, W; Altman, MD; Dahlberg, WK; Davis, L; Falcone, D; Gabarda, AE; Hang, G; Hatch, H; Holmes, R; Kunii, K; Lumb, KJ; Lutterbach, B; Mathvink, R; Nazef, N; Patel, SB; Qu, X; Reilly, JF; Rickert, KW; Rosenstein, C; Soisson, SM; Spencer, KB; Szewczak, AA; Walker, D; Wang, W; Young, J; Zeng, Q Discovery of a 5H-benzo[4,5]cyclohepta[1,2-b]pyridin-5-one (MK-2461) inhibitor of c-Met kinase for the treatment of cancer. J Med Chem54:4092-108 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50347656 |
|---|
| n/a |
|---|
| Name | BDBM50347656 |
|---|
| Synonyms: | CHEMBL1803004 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H16N2O3S |
|---|
| Mol. Mass. | 376.428 |
|---|
| SMILES | CS(=O)(=O)Nc1ccc2ccc3ncc(cc3c(=O)c2c1)-c1ccccc1 |
|---|
| Structure | 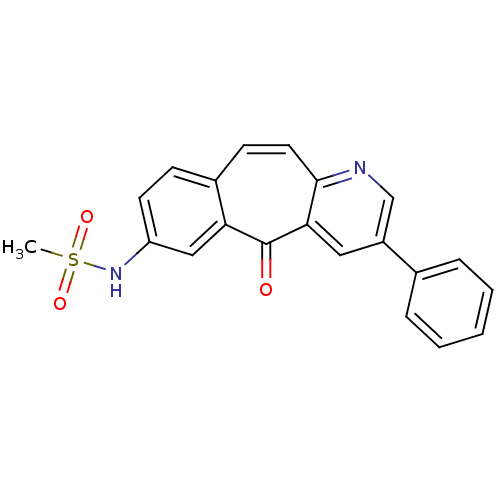
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Katz, JD; Jewell, JP; Guerin, DJ; Lim, J; Dinsmore, CJ; Deshmukh, SV; Pan, BS; Marshall, CG; Lu, W; Altman, MD; Dahlberg, WK; Davis, L; Falcone, D; Gabarda, AE; Hang, G; Hatch, H; Holmes, R; Kunii, K; Lumb, KJ; Lutterbach, B; Mathvink, R; Nazef, N; Patel, SB; Qu, X; Reilly, JF; Rickert, KW; Rosenstein, C; Soisson, SM; Spencer, KB; Szewczak, AA; Walker, D; Wang, W; Young, J; Zeng, Q Discovery of a 5H-benzo[4,5]cyclohepta[1,2-b]pyridin-5-one (MK-2461) inhibitor of c-Met kinase for the treatment of cancer. J Med Chem54:4092-108 (2011) [PubMed] Article
Katz, JD; Jewell, JP; Guerin, DJ; Lim, J; Dinsmore, CJ; Deshmukh, SV; Pan, BS; Marshall, CG; Lu, W; Altman, MD; Dahlberg, WK; Davis, L; Falcone, D; Gabarda, AE; Hang, G; Hatch, H; Holmes, R; Kunii, K; Lumb, KJ; Lutterbach, B; Mathvink, R; Nazef, N; Patel, SB; Qu, X; Reilly, JF; Rickert, KW; Rosenstein, C; Soisson, SM; Spencer, KB; Szewczak, AA; Walker, D; Wang, W; Young, J; Zeng, Q Discovery of a 5H-benzo[4,5]cyclohepta[1,2-b]pyridin-5-one (MK-2461) inhibitor of c-Met kinase for the treatment of cancer. J Med Chem54:4092-108 (2011) [PubMed] Article