| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50379186 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_810646 (CHEMBL2015697) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Hudkins, RL; Becknell, NC; Zulli, AL; Underiner, TL; Angeles, TS; Aimone, LD; Albom, MS; Chang, H; Miknyoczki, SJ; Hunter, K; Jones-Bolin, S; Zhao, H; Bacon, ER; Mallamo, JP; Ator, MA; Ruggeri, BA Synthesis and biological profile of the pan-vascular endothelial growth factor receptor/tyrosine kinase with immunoglobulin and epidermal growth factor-like homology domains 2 (VEGF-R/TIE-2) inhibitor 11-(2-methylpropyl)-12,13-dihydro-2-methyl-8-(pyrimidin-2-ylamino)-4H-indazolo[5,4-a]pyrrolo[3,4-c J Med Chem55:903-13 (2012) [PubMed] Article Hudkins, RL; Becknell, NC; Zulli, AL; Underiner, TL; Angeles, TS; Aimone, LD; Albom, MS; Chang, H; Miknyoczki, SJ; Hunter, K; Jones-Bolin, S; Zhao, H; Bacon, ER; Mallamo, JP; Ator, MA; Ruggeri, BA Synthesis and biological profile of the pan-vascular endothelial growth factor receptor/tyrosine kinase with immunoglobulin and epidermal growth factor-like homology domains 2 (VEGF-R/TIE-2) inhibitor 11-(2-methylpropyl)-12,13-dihydro-2-methyl-8-(pyrimidin-2-ylamino)-4H-indazolo[5,4-a]pyrrolo[3,4-c J Med Chem55:903-13 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50379186 |
|---|
| n/a |
|---|
| Name | BDBM50379186 |
|---|
| Synonyms: | CEP-11981 | CHEMBL2010872 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H27N7O |
|---|
| Mol. Mass. | 477.5603 |
|---|
| SMILES | CC(C)Cn1c2ccc(Nc3ncccn3)cc2c2c3CNC(=O)c3c3-c4cn(C)nc4CCc3c12 |
|---|
| Structure | 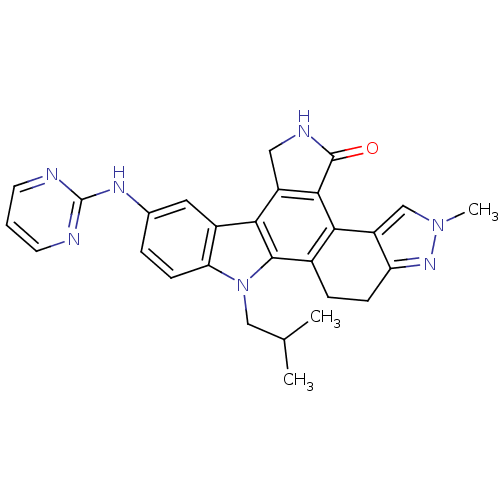
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hudkins, RL; Becknell, NC; Zulli, AL; Underiner, TL; Angeles, TS; Aimone, LD; Albom, MS; Chang, H; Miknyoczki, SJ; Hunter, K; Jones-Bolin, S; Zhao, H; Bacon, ER; Mallamo, JP; Ator, MA; Ruggeri, BA Synthesis and biological profile of the pan-vascular endothelial growth factor receptor/tyrosine kinase with immunoglobulin and epidermal growth factor-like homology domains 2 (VEGF-R/TIE-2) inhibitor 11-(2-methylpropyl)-12,13-dihydro-2-methyl-8-(pyrimidin-2-ylamino)-4H-indazolo[5,4-a]pyrrolo[3,4-c J Med Chem55:903-13 (2012) [PubMed] Article
Hudkins, RL; Becknell, NC; Zulli, AL; Underiner, TL; Angeles, TS; Aimone, LD; Albom, MS; Chang, H; Miknyoczki, SJ; Hunter, K; Jones-Bolin, S; Zhao, H; Bacon, ER; Mallamo, JP; Ator, MA; Ruggeri, BA Synthesis and biological profile of the pan-vascular endothelial growth factor receptor/tyrosine kinase with immunoglobulin and epidermal growth factor-like homology domains 2 (VEGF-R/TIE-2) inhibitor 11-(2-methylpropyl)-12,13-dihydro-2-methyl-8-(pyrimidin-2-ylamino)-4H-indazolo[5,4-a]pyrrolo[3,4-c J Med Chem55:903-13 (2012) [PubMed] Article