| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50380551 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_813114 (CHEMBL2020733) |
|---|
| IC50 | 0.10±n/a nM |
|---|
| Citation |  Fernández-Bachiller, MI; Pérez, C; Monjas, L; Rademann, J; Rodríguez-Franco, MI New tacrine-4-oxo-4H-chromene hybrids as multifunctional agents for the treatment of Alzheimer's disease, with cholinergic, antioxidant, andß-amyloid-reducing properties. J Med Chem55:1303-17 (2012) [PubMed] Article Fernández-Bachiller, MI; Pérez, C; Monjas, L; Rademann, J; Rodríguez-Franco, MI New tacrine-4-oxo-4H-chromene hybrids as multifunctional agents for the treatment of Alzheimer's disease, with cholinergic, antioxidant, andß-amyloid-reducing properties. J Med Chem55:1303-17 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | Acylcholine acylhydrolase | BCHE | Butyrylcholine esterase (BChE) | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | CHE1 | CHLE_HUMAN | Choline esterase II | Cholinesterases | Cholinesterases; ACHE & BCHE | Pseudocholinesterase |
|---|
| Type: | Homotetramer |
|---|
| Mol. Mass.: | 68422.27 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06276 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MHSKVTIICIRFLFWFLLLCMLIGKSHTEDDIIIATKNGKVRGMNLTVFGGTVTAFLGIP
YAQPPLGRLRFKKPQSLTKWSDIWNATKYANSCCQNIDQSFPGFHGSEMWNPNTDLSEDC
LYLNVWIPAPKPKNATVLIWIYGGGFQTGTSSLHVYDGKFLARVERVIVVSMNYRVGALG
FLALPGNPEAPGNMGLFDQQLALQWVQKNIAAFGGNPKSVTLFGESAGAASVSLHLLSPG
SHSLFTRAILQSGSFNAPWAVTSLYEARNRTLNLAKLTGCSRENETEIIKCLRNKDPQEI
LLNEAFVVPYGTPLSVNFGPTVDGDFLTDMPDILLELGQFKKTQILVGVNKDEGTAFLVY
GAPGFSKDNNSIITRKEFQEGLKIFFPGVSEFGKESILFHYTDWVDDQRPENYREALGDV
VGDYNFICPALEFTKKFSEWGNNAFFYYFEHRSSKLPWPEWMGVMHGYEIEFVFGLPLER
RDNYTKAEEILSRSIVKRWANFAKYGNPNETQNNSTSWPVFKSTEQKYLTLNTESTRIMT
KLRAQQCRFWTSFFPKVLEMTGNIDEAEWEWKAGFHRWNNYMMDWKNQFNDYTSKKESCV
GL
|
|
|
|---|
| BDBM50380551 |
|---|
| n/a |
|---|
| Name | BDBM50380551 |
|---|
| Synonyms: | CHEMBL2019038 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C35H42ClN3O5 |
|---|
| Mol. Mass. | 620.178 |
|---|
| SMILES | COc1cc(OC)c2c(c1)oc(cc2=O)C(=O)NCCCCCCCCCCNc1c2CCCCc2nc2cc(Cl)ccc12 |
|---|
| Structure | 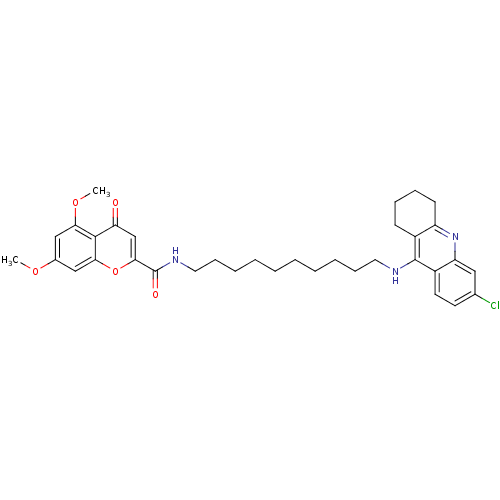
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fernández-Bachiller, MI; Pérez, C; Monjas, L; Rademann, J; Rodríguez-Franco, MI New tacrine-4-oxo-4H-chromene hybrids as multifunctional agents for the treatment of Alzheimer's disease, with cholinergic, antioxidant, andß-amyloid-reducing properties. J Med Chem55:1303-17 (2012) [PubMed] Article
Fernández-Bachiller, MI; Pérez, C; Monjas, L; Rademann, J; Rodríguez-Franco, MI New tacrine-4-oxo-4H-chromene hybrids as multifunctional agents for the treatment of Alzheimer's disease, with cholinergic, antioxidant, andß-amyloid-reducing properties. J Med Chem55:1303-17 (2012) [PubMed] Article