| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50381716 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_816384 (CHEMBL2024782) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Curtin, ML; Frey, RR; Heyman, HR; Soni, NB; Marcotte, PA; Pease, LJ; Glaser, KB; Magoc, TJ; Tapang, P; Albert, DH; Osterling, DJ; Olson, AM; Bouska, JJ; Guan, Z; Preusser, LC; Polakowski, JS; Stewart, KD; Tse, C; Davidsen, SK; Michaelides, MR Thienopyridine ureas as dual inhibitors of the VEGF and Aurora kinase families. Bioorg Med Chem Lett22:3208-12 (2012) [PubMed] Article Curtin, ML; Frey, RR; Heyman, HR; Soni, NB; Marcotte, PA; Pease, LJ; Glaser, KB; Magoc, TJ; Tapang, P; Albert, DH; Osterling, DJ; Olson, AM; Bouska, JJ; Guan, Z; Preusser, LC; Polakowski, JS; Stewart, KD; Tse, C; Davidsen, SK; Michaelides, MR Thienopyridine ureas as dual inhibitors of the VEGF and Aurora kinase families. Bioorg Med Chem Lett22:3208-12 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50381716 |
|---|
| n/a |
|---|
| Name | BDBM50381716 |
|---|
| Synonyms: | ABT-348 | ILORASERTIB | US8722890, 1 | US8722890, 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H21FN6O2S |
|---|
| Mol. Mass. | 488.537 |
|---|
| SMILES | Nc1ncc(-c2cnn(CCO)c2)c2scc(-c3ccc(NC(=O)Nc4cccc(F)c4)cc3)c12 |
|---|
| Structure | 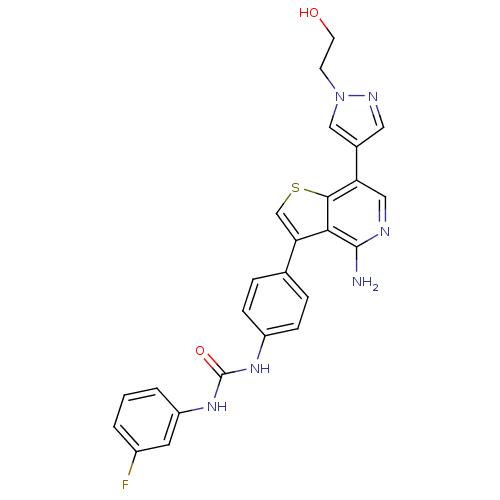
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Curtin, ML; Frey, RR; Heyman, HR; Soni, NB; Marcotte, PA; Pease, LJ; Glaser, KB; Magoc, TJ; Tapang, P; Albert, DH; Osterling, DJ; Olson, AM; Bouska, JJ; Guan, Z; Preusser, LC; Polakowski, JS; Stewart, KD; Tse, C; Davidsen, SK; Michaelides, MR Thienopyridine ureas as dual inhibitors of the VEGF and Aurora kinase families. Bioorg Med Chem Lett22:3208-12 (2012) [PubMed] Article
Curtin, ML; Frey, RR; Heyman, HR; Soni, NB; Marcotte, PA; Pease, LJ; Glaser, KB; Magoc, TJ; Tapang, P; Albert, DH; Osterling, DJ; Olson, AM; Bouska, JJ; Guan, Z; Preusser, LC; Polakowski, JS; Stewart, KD; Tse, C; Davidsen, SK; Michaelides, MR Thienopyridine ureas as dual inhibitors of the VEGF and Aurora kinase families. Bioorg Med Chem Lett22:3208-12 (2012) [PubMed] Article