| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50385246 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_822402 (CHEMBL2038950) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Becknell, NC; Lyons, JA; Aimone, LD; Huang, Z; Gruner, JA; Raddatz, R; Hudkins, RL Synthesis and evaluation of 4- and 5-pyridazin-3-one phenoxypropylamine analogues as histamine-3 receptor antagonists. Bioorg Med Chem20:3880-6 (2012) [PubMed] Article Becknell, NC; Lyons, JA; Aimone, LD; Huang, Z; Gruner, JA; Raddatz, R; Hudkins, RL Synthesis and evaluation of 4- and 5-pyridazin-3-one phenoxypropylamine analogues as histamine-3 receptor antagonists. Bioorg Med Chem20:3880-6 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50385246 |
|---|
| n/a |
|---|
| Name | BDBM50385246 |
|---|
| Synonyms: | CHEMBL2037604 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H25N3O2 |
|---|
| Mol. Mass. | 327.4207 |
|---|
| SMILES | C[C@H](COc1ccc(cc1)-c1cn[nH]c(=O)c1)CN1CCC[C@H]1C |r| |
|---|
| Structure | 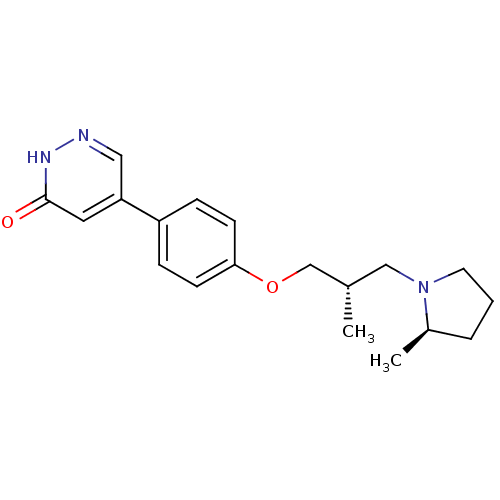
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Becknell, NC; Lyons, JA; Aimone, LD; Huang, Z; Gruner, JA; Raddatz, R; Hudkins, RL Synthesis and evaluation of 4- and 5-pyridazin-3-one phenoxypropylamine analogues as histamine-3 receptor antagonists. Bioorg Med Chem20:3880-6 (2012) [PubMed] Article
Becknell, NC; Lyons, JA; Aimone, LD; Huang, Z; Gruner, JA; Raddatz, R; Hudkins, RL Synthesis and evaluation of 4- and 5-pyridazin-3-one phenoxypropylamine analogues as histamine-3 receptor antagonists. Bioorg Med Chem20:3880-6 (2012) [PubMed] Article