| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M1 |
|---|
| Ligand | BDBM50165019 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_826078 (CHEMBL2045509) |
|---|
| Ki | 2.4±n/a nM |
|---|
| Citation |  Del Bello, F; Barocelli, E; Bertoni, S; Bonifazi, A; Camalli, M; Campi, G; Giannella, M; Matucci, R; Nesi, M; Pigini, M; Quaglia, W; Piergentili, A 1,4-dioxane, a suitable scaffold for the development of novel M3 muscarinic receptor antagonists. J Med Chem55:1783-7 (2012) [PubMed] Article Del Bello, F; Barocelli, E; Bertoni, S; Bonifazi, A; Camalli, M; Campi, G; Giannella, M; Matucci, R; Nesi, M; Pigini, M; Quaglia, W; Piergentili, A 1,4-dioxane, a suitable scaffold for the development of novel M3 muscarinic receptor antagonists. J Med Chem55:1783-7 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M1 |
|---|
| Name: | Muscarinic acetylcholine receptor M1 |
|---|
| Synonyms: | ACM1_HUMAN | CHRM1 | Cholinergic receptor, muscarinic 1 | Cholinergic, muscarinic M1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 51442.54 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11229 |
|---|
| Residue: | 460 |
|---|
| Sequence: | MNTSAPPAVSPNITVLAPGKGPWQVAFIGITTGLLSLATVTGNLLVLISFKVNTELKTVN
NYFLLSLACADLIIGTFSMNLYTTYLLMGHWALGTLACDLWLALDYVASNASVMNLLLIS
FDRYFSVTRPLSYRAKRTPRRAALMIGLAWLVSFVLWAPAILFWQYLVGERTVLAGQCYI
QFLSQPIITFGTAMAAFYLPVTVMCTLYWRIYRETENRARELAALQGSETPGKGGGSSSS
SERSQPGAEGSPETPPGRCCRCCRAPRLLQAYSWKEEEEEDEGSMESLTSSEGEEPGSEV
VIKMPMVDPEAQAPTKQPPRSSPNTVKRPTKKGRDRAGKGQKPRGKEQLAKRKTFSLVKE
KKAARTLSAILLAFILTWTPYNIMVLVSTFCKDCVPETLWELGYWLCYVNSTINPMCYAL
CNKAFRDTFRLLLLCRWDKRRWRKIPKRPGSVHRTPSRQC
|
|
|
|---|
| BDBM50165019 |
|---|
| n/a |
|---|
| Name | BDBM50165019 |
|---|
| Synonyms: | 4-(Diethylamino)-2-butynyl alpha-phenylcyclohexaneglycolic acid ester | 4-(diethylamino)but-2-yn-1-yl cyclohexyl(hydroxy)phenylacetate | 4-Diethylamino-2-butinyl alpha-cyclohexylmandelat | 4-Diethylamino-2-butynyl alpha-phenylcyclohexaneglycolate | Benzeneacetic acid, alpha-cyclohexyl-alpha-hydroxy-, 4-(diethylamino)-2-butynyl ester | CHEMBL1231 | Cyclohexaneglycolic acid, alpha-phenyl-, 4-(diethylamino)-2-butynyl ester | OXYBUTYNIN CHLORIDE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H31NO3 |
|---|
| Mol. Mass. | 357.4864 |
|---|
| SMILES | CCN(CC)CC#CCOC(=O)C(O)(C1CCCCC1)c1ccccc1 |
|---|
| Structure | 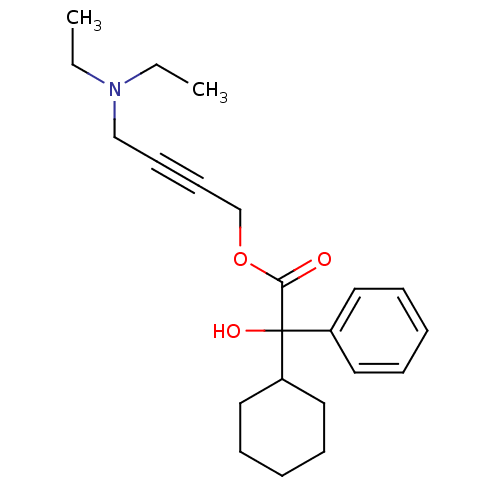
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Del Bello, F; Barocelli, E; Bertoni, S; Bonifazi, A; Camalli, M; Campi, G; Giannella, M; Matucci, R; Nesi, M; Pigini, M; Quaglia, W; Piergentili, A 1,4-dioxane, a suitable scaffold for the development of novel M3 muscarinic receptor antagonists. J Med Chem55:1783-7 (2012) [PubMed] Article
Del Bello, F; Barocelli, E; Bertoni, S; Bonifazi, A; Camalli, M; Campi, G; Giannella, M; Matucci, R; Nesi, M; Pigini, M; Quaglia, W; Piergentili, A 1,4-dioxane, a suitable scaffold for the development of novel M3 muscarinic receptor antagonists. J Med Chem55:1783-7 (2012) [PubMed] Article