| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50385814 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_823568 (CHEMBL2046011) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Vachal, P; Miao, S; Pierce, JM; Guiadeen, D; Colandrea, VJ; Wyvratt, MJ; Salowe, SP; Sonatore, LM; Milligan, JA; Hajdu, R; Gollapudi, A; Keohane, CA; Lingham, RB; Mandala, SM; DeMartino, JA; Tong, X; Wolff, M; Steinhuebel, D; Kieczykowski, GR; Fleitz, FJ; Chapman, K; Athanasopoulos, J; Adam, G; Akyuz, CD; Jena, DK; Lusen, JW; Meng, J; Stein, BD; Xia, L; Sherer, EC; Hale, JJ 1,3,8-Triazaspiro[4.5]decane-2,4-diones as efficacious pan-inhibitors of hypoxia-inducible factor prolyl hydroxylase 1-3 (HIF PHD1-3) for the treatment of anemia. J Med Chem55:2945-59 (2012) [PubMed] Article Vachal, P; Miao, S; Pierce, JM; Guiadeen, D; Colandrea, VJ; Wyvratt, MJ; Salowe, SP; Sonatore, LM; Milligan, JA; Hajdu, R; Gollapudi, A; Keohane, CA; Lingham, RB; Mandala, SM; DeMartino, JA; Tong, X; Wolff, M; Steinhuebel, D; Kieczykowski, GR; Fleitz, FJ; Chapman, K; Athanasopoulos, J; Adam, G; Akyuz, CD; Jena, DK; Lusen, JW; Meng, J; Stein, BD; Xia, L; Sherer, EC; Hale, JJ 1,3,8-Triazaspiro[4.5]decane-2,4-diones as efficacious pan-inhibitors of hypoxia-inducible factor prolyl hydroxylase 1-3 (HIF PHD1-3) for the treatment of anemia. J Med Chem55:2945-59 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50385814 |
|---|
| n/a |
|---|
| Name | BDBM50385814 |
|---|
| Synonyms: | CHEMBL2043169 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H30N6O5 |
|---|
| Mol. Mass. | 578.6178 |
|---|
| SMILES | COc1cc(ncn1)N1C(=O)N(C(=O)C11CCN(Cc2ncccc2C)CC1)c1ccc(cc1)-c1cccc(c1)C(O)=O |
|---|
| Structure | 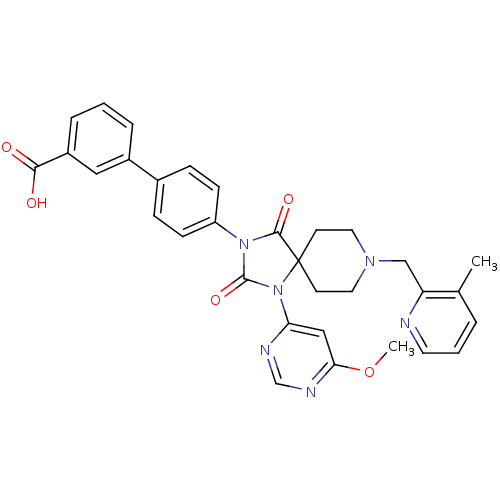
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Vachal, P; Miao, S; Pierce, JM; Guiadeen, D; Colandrea, VJ; Wyvratt, MJ; Salowe, SP; Sonatore, LM; Milligan, JA; Hajdu, R; Gollapudi, A; Keohane, CA; Lingham, RB; Mandala, SM; DeMartino, JA; Tong, X; Wolff, M; Steinhuebel, D; Kieczykowski, GR; Fleitz, FJ; Chapman, K; Athanasopoulos, J; Adam, G; Akyuz, CD; Jena, DK; Lusen, JW; Meng, J; Stein, BD; Xia, L; Sherer, EC; Hale, JJ 1,3,8-Triazaspiro[4.5]decane-2,4-diones as efficacious pan-inhibitors of hypoxia-inducible factor prolyl hydroxylase 1-3 (HIF PHD1-3) for the treatment of anemia. J Med Chem55:2945-59 (2012) [PubMed] Article
Vachal, P; Miao, S; Pierce, JM; Guiadeen, D; Colandrea, VJ; Wyvratt, MJ; Salowe, SP; Sonatore, LM; Milligan, JA; Hajdu, R; Gollapudi, A; Keohane, CA; Lingham, RB; Mandala, SM; DeMartino, JA; Tong, X; Wolff, M; Steinhuebel, D; Kieczykowski, GR; Fleitz, FJ; Chapman, K; Athanasopoulos, J; Adam, G; Akyuz, CD; Jena, DK; Lusen, JW; Meng, J; Stein, BD; Xia, L; Sherer, EC; Hale, JJ 1,3,8-Triazaspiro[4.5]decane-2,4-diones as efficacious pan-inhibitors of hypoxia-inducible factor prolyl hydroxylase 1-3 (HIF PHD1-3) for the treatment of anemia. J Med Chem55:2945-59 (2012) [PubMed] Article