| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor |
|---|
| Ligand | BDBM50262235 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_828757 (CHEMBL2060407) |
|---|
| Ki | 36±n/a nM |
|---|
| Citation |  Schann, S; Greney, H; Gasparik, V; Dontenwill, M; Rascente, C; Lacroix, G; Monassier, L; Bruban, V; Feldman, J; Ehrhardt, JD; Bousquet, P Methylation of imidazoline related compounds leads to loss ofa2-adrenoceptor affinity. Synthesis and biological evaluation of selective I1 imidazoline receptor ligands. Bioorg Med Chem20:4710-5 (2012) [PubMed] Article Schann, S; Greney, H; Gasparik, V; Dontenwill, M; Rascente, C; Lacroix, G; Monassier, L; Bruban, V; Feldman, J; Ehrhardt, JD; Bousquet, P Methylation of imidazoline related compounds leads to loss ofa2-adrenoceptor affinity. Synthesis and biological evaluation of selective I1 imidazoline receptor ligands. Bioorg Med Chem20:4710-5 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2A adrenergic receptor |
|---|
| Name: | Alpha-2A adrenergic receptor |
|---|
| Synonyms: | ADA2A_HUMAN | ADRA2A | ADRA2R | ADRAR | Adrenergic alpha2A | Adrenergic receptor alpha | Alpha-2 adrenergic receptor subtype C10 | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | alpha-2A adrenergic receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48979.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08913 |
|---|
| Residue: | 450 |
|---|
| Sequence: | MGSLQPDAGNASWNGTEAPGGGARATPYSLQVTLTLVCLAGLLMLLTVFGNVLVIIAVFT
SRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKAWCEIYLALDVLFCTSS
IVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIITVWVISAVISFPPLISIEKKGGGGG
PQPAEPRCEINDQKWYVISSCIGSFFAPCLIMILVYVRIYQIAKRRTRVPPSRRGPDAVA
APPGGTERRPNGLGPERSAGPGGAEAEPLPTQLNGAPGEPAPAGPRDTDALDLEESSSSD
HAERPPGPRRPERGPRGKGKARASQVKPGDSLPRRGPGATGIGTPAAGPGEERVGAAKAS
RWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLTAVGCSVPRTLFKFFFWFGYCNSS
LNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM50262235 |
|---|
| n/a |
|---|
| Name | BDBM50262235 |
|---|
| Synonyms: | rilmenidine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H17N2O |
|---|
| Mol. Mass. | 181.2542 |
|---|
| SMILES | C1CC1C(NC1=[NH+]CCO1)C1CC1 |t:6| |
|---|
| Structure | 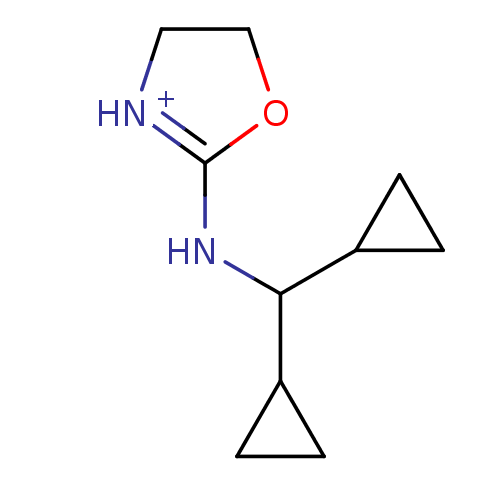
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Schann, S; Greney, H; Gasparik, V; Dontenwill, M; Rascente, C; Lacroix, G; Monassier, L; Bruban, V; Feldman, J; Ehrhardt, JD; Bousquet, P Methylation of imidazoline related compounds leads to loss ofa2-adrenoceptor affinity. Synthesis and biological evaluation of selective I1 imidazoline receptor ligands. Bioorg Med Chem20:4710-5 (2012) [PubMed] Article
Schann, S; Greney, H; Gasparik, V; Dontenwill, M; Rascente, C; Lacroix, G; Monassier, L; Bruban, V; Feldman, J; Ehrhardt, JD; Bousquet, P Methylation of imidazoline related compounds leads to loss ofa2-adrenoceptor affinity. Synthesis and biological evaluation of selective I1 imidazoline receptor ligands. Bioorg Med Chem20:4710-5 (2012) [PubMed] Article