| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Endothelin-1 receptor |
|---|
| Ligand | BDBM50395665 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_859881 (CHEMBL2166382) |
|---|
| IC50 | 2.8±n/a nM |
|---|
| Citation |  Bolli, MH; Boss, C; Binkert, C; Buchmann, S; Bur, D; Hess, P; Iglarz, M; Meyer, S; Rein, J; Rey, M; Treiber, A; Clozel, M; Fischli, W; Weller, T The discovery of N-[5-(4-bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide (Macitentan), an orally active, potent dual endothelin receptor antagonist. J Med Chem55:7849-61 (2012) [PubMed] Article Bolli, MH; Boss, C; Binkert, C; Buchmann, S; Bur, D; Hess, P; Iglarz, M; Meyer, S; Rein, J; Rey, M; Treiber, A; Clozel, M; Fischli, W; Weller, T The discovery of N-[5-(4-bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide (Macitentan), an orally active, potent dual endothelin receptor antagonist. J Med Chem55:7849-61 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Endothelin-1 receptor |
|---|
| Name: | Endothelin-1 receptor |
|---|
| Synonyms: | EDNRA | EDNRA_HUMAN | ET-A | ETA | ETA-R | ETRA | Endothelin receptor type A | Endothelin receptor, ET-A/ET-B | hET-AR |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 48736.88 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25101 |
|---|
| Residue: | 427 |
|---|
| Sequence: | METLCLRASFWLALVGCVISDNPERYSTNLSNHVDDFTTFRGTELSFLVTTHQPTNLVLP
SNGSMHNYCPQQTKITSAFKYINTVISCTIFIVGMVGNATLLRIIYQNKCMRNGPNALIA
SLALGDLIYVVIDLPINVFKLLAGRWPFDHNDFGVFLCKLFPFLQKSSVGITVLNLCALS
VDRYRAVASWSRVQGIGIPLVTAIEIVSIWILSFILAIPEAIGFVMVPFEYRGEQHKTCM
LNATSKFMEFYQDVKDWWLFGFYFCMPLVCTAIFYTLMTCEMLNRRNGSLRIALSEHLKQ
RREVAKTVFCLVVIFALCWFPLHLSRILKKTVYNEMDKNRCELLSFLLLMDYIGINLATM
NSCINPIALYFVSKKFKNCFQSCLCCCCYQSKSLMTSVPMNGTSIQWKNHDQNNHNTDRS
SHKDSMN
|
|
|
|---|
| BDBM50395665 |
|---|
| n/a |
|---|
| Name | BDBM50395665 |
|---|
| Synonyms: | CHEMBL2163701 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H26ClN7O6S |
|---|
| Mol. Mass. | 636.078 |
|---|
| SMILES | COc1ccccc1Oc1c(NS(=O)(=O)CCc2ccccc2)nc(nc1OCCOc1ncc(Cl)cn1)-c1ncccn1 |
|---|
| Structure | 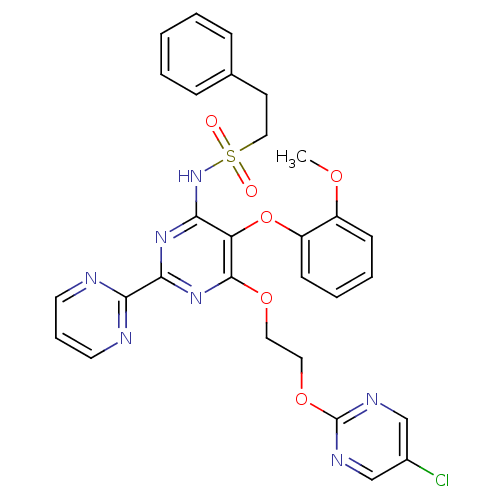
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bolli, MH; Boss, C; Binkert, C; Buchmann, S; Bur, D; Hess, P; Iglarz, M; Meyer, S; Rein, J; Rey, M; Treiber, A; Clozel, M; Fischli, W; Weller, T The discovery of N-[5-(4-bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide (Macitentan), an orally active, potent dual endothelin receptor antagonist. J Med Chem55:7849-61 (2012) [PubMed] Article
Bolli, MH; Boss, C; Binkert, C; Buchmann, S; Bur, D; Hess, P; Iglarz, M; Meyer, S; Rein, J; Rey, M; Treiber, A; Clozel, M; Fischli, W; Weller, T The discovery of N-[5-(4-bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide (Macitentan), an orally active, potent dual endothelin receptor antagonist. J Med Chem55:7849-61 (2012) [PubMed] Article