| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin K |
|---|
| Ligand | BDBM19855 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_864178 (CHEMBL2175127) |
|---|
| IC50 | 5±n/a nM |
|---|
| Citation |  Crawford, JJ; Kenny, PW; Bowyer, J; Cook, CR; Finlayson, JE; Heyes, C; Highton, AJ; Hudson, JA; Jestel, A; Krapp, S; Martin, S; Macfaul, PA; McDermott, BP; McGuire, TM; Morley, AD; Morris, JJ; Page, KM; Ribeiro, LR; Sawney, H; Steinbacher, S; Smith, C; Dossetter, AG Pharmacokinetic benefits of 3,4-dimethoxy substitution of a phenyl ring and design of isosteres yielding orally available cathepsin K inhibitors. J Med Chem55:8827-37 (2012) [PubMed] Article Crawford, JJ; Kenny, PW; Bowyer, J; Cook, CR; Finlayson, JE; Heyes, C; Highton, AJ; Hudson, JA; Jestel, A; Krapp, S; Martin, S; Macfaul, PA; McDermott, BP; McGuire, TM; Morley, AD; Morris, JJ; Page, KM; Ribeiro, LR; Sawney, H; Steinbacher, S; Smith, C; Dossetter, AG Pharmacokinetic benefits of 3,4-dimethoxy substitution of a phenyl ring and design of isosteres yielding orally available cathepsin K inhibitors. J Med Chem55:8827-37 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin K |
|---|
| Name: | Cathepsin K |
|---|
| Synonyms: | CATK_HUMAN | CTSK | CTSO | CTSO2 | Cathepsin O | Cathepsin O2 | Cathepsin X |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36975.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P43235 |
|---|
| Residue: | 329 |
|---|
| Sequence: | MWGLKVLLLPVVSFALYPEEILDTHWELWKKTHRKQYNNKVDEISRRLIWEKNLKYISIH
NLEASLGVHTYELAMNHLGDMTSEEVVQKMTGLKVPLSHSRSNDTLYIPEWEGRAPDSVD
YRKKGYVTPVKNQGQCGSCWAFSSVGALEGQLKKKTGKLLNLSPQNLVDCVSENDGCGGG
YMTNAFQYVQKNRGIDSEDAYPYVGQEESCMYNPTGKAAKCRGYREIPEGNEKALKRAVA
RVGPVSVAIDASLTSFQFYSKGVYYDESCNSDNLNHAVLAVGYGIQKGNKHWIIKNSWGE
NWGNKGYILMARNKNNACGIANLASFPKM
|
|
|
|---|
| BDBM19855 |
|---|
| n/a |
|---|
| Name | BDBM19855 |
|---|
| Synonyms: | Balicatib | CHEMBL371064 | N-[1-(cyanomethylcarbamoyl)cyclohexyl]-4-(4-propylpiperazin-1-yl) | N-{1-[(cyanomethyl)carbamoyl]cyclohexyl}-4-(4-propylpiperazin-1-yl)benzamide | basic piperazine-containing compound, 10 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H33N5O2 |
|---|
| Mol. Mass. | 411.5404 |
|---|
| SMILES | CCCN1CCN(CC1)c1ccc(cc1)C(=O)NC1(CCCCC1)C(=O)NCC#N |
|---|
| Structure | 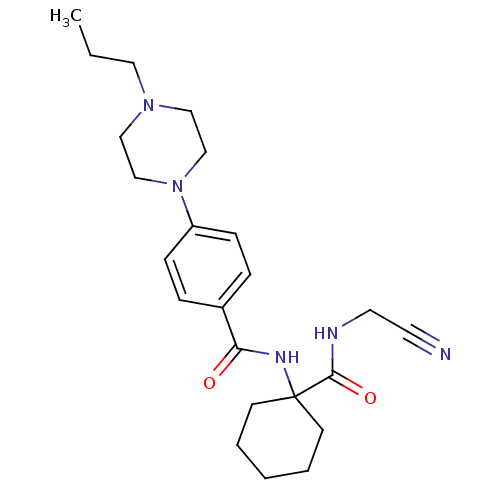
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Crawford, JJ; Kenny, PW; Bowyer, J; Cook, CR; Finlayson, JE; Heyes, C; Highton, AJ; Hudson, JA; Jestel, A; Krapp, S; Martin, S; Macfaul, PA; McDermott, BP; McGuire, TM; Morley, AD; Morris, JJ; Page, KM; Ribeiro, LR; Sawney, H; Steinbacher, S; Smith, C; Dossetter, AG Pharmacokinetic benefits of 3,4-dimethoxy substitution of a phenyl ring and design of isosteres yielding orally available cathepsin K inhibitors. J Med Chem55:8827-37 (2012) [PubMed] Article
Crawford, JJ; Kenny, PW; Bowyer, J; Cook, CR; Finlayson, JE; Heyes, C; Highton, AJ; Hudson, JA; Jestel, A; Krapp, S; Martin, S; Macfaul, PA; McDermott, BP; McGuire, TM; Morley, AD; Morris, JJ; Page, KM; Ribeiro, LR; Sawney, H; Steinbacher, S; Smith, C; Dossetter, AG Pharmacokinetic benefits of 3,4-dimethoxy substitution of a phenyl ring and design of isosteres yielding orally available cathepsin K inhibitors. J Med Chem55:8827-37 (2012) [PubMed] Article