| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50407259 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_43176 (CHEMBL659203) |
|---|
| EC50 | 280±n/a nM |
|---|
| Citation |  Kumar, R; Wang, L; Wiebe, LI; Knaus, EE Synthesis, in vitro biological stability, and anti-HIV activity of 5-halo-6-alkoxy(or azido)-5,6-dihydro-3'-azido-3'-deoxythymidine diastereomers as potential prodrugs to 3'-azido-3'-deoxythymidine (AZT). J Med Chem37:4297-306 (1995) [PubMed] Kumar, R; Wang, L; Wiebe, LI; Knaus, EE Synthesis, in vitro biological stability, and anti-HIV activity of 5-halo-6-alkoxy(or azido)-5,6-dihydro-3'-azido-3'-deoxythymidine diastereomers as potential prodrugs to 3'-azido-3'-deoxythymidine (AZT). J Med Chem37:4297-306 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50407259 |
|---|
| n/a |
|---|
| Name | BDBM50407259 |
|---|
| Synonyms: | CHEMBL342041 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H16BrN5O5 |
|---|
| Mol. Mass. | 378.179 |
|---|
| SMILES | CO[C@@H]1N(C2CC(N=[N+]=[N-])C(CO)O2)C(=O)NC(=O)[C@@]1(C)Br |
|---|
| Structure | 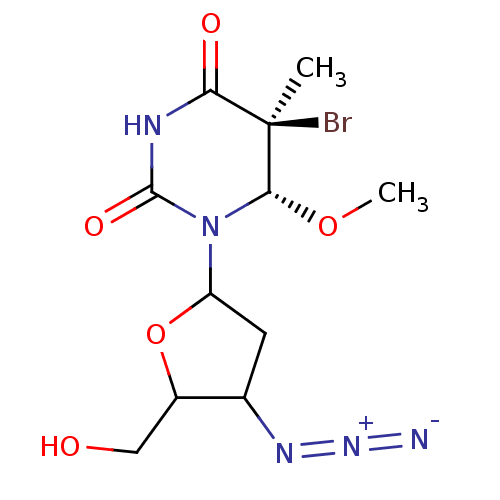
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kumar, R; Wang, L; Wiebe, LI; Knaus, EE Synthesis, in vitro biological stability, and anti-HIV activity of 5-halo-6-alkoxy(or azido)-5,6-dihydro-3'-azido-3'-deoxythymidine diastereomers as potential prodrugs to 3'-azido-3'-deoxythymidine (AZT). J Med Chem37:4297-306 (1995) [PubMed]
Kumar, R; Wang, L; Wiebe, LI; Knaus, EE Synthesis, in vitro biological stability, and anti-HIV activity of 5-halo-6-alkoxy(or azido)-5,6-dihydro-3'-azido-3'-deoxythymidine diastereomers as potential prodrugs to 3'-azido-3'-deoxythymidine (AZT). J Med Chem37:4297-306 (1995) [PubMed]