| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calcitonin gene-related peptide type 1 receptor |
|---|
| Ligand | BDBM50415360 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_608969 (CHEMBL1073111) |
|---|
| Ki | 3.16±n/a nM |
|---|
| Citation |  Nichols, PL; Brand, J; Briggs, M; D'Angeli, M; Farge, J; Garland, SL; Goldsmith, P; Hutchings, R; Kilford, I; Li, HY; MacPherson, D; Nimmo, F; Sanderson, FD; Sehmi, S; Shuker, N; Skidmore, J; Stott, M; Sweeting, J; Tajuddin, H; Takle, AK; Trani, G; Wall, ID; Ward, R; Wilson, DM; Witty, D Potent oxadiazole CGRP receptor antagonists for the potential treatment of migraine. Bioorg Med Chem Lett20:1368-72 (2010) [PubMed] Article Nichols, PL; Brand, J; Briggs, M; D'Angeli, M; Farge, J; Garland, SL; Goldsmith, P; Hutchings, R; Kilford, I; Li, HY; MacPherson, D; Nimmo, F; Sanderson, FD; Sehmi, S; Shuker, N; Skidmore, J; Stott, M; Sweeting, J; Tajuddin, H; Takle, AK; Trani, G; Wall, ID; Ward, R; Wilson, DM; Witty, D Potent oxadiazole CGRP receptor antagonists for the potential treatment of migraine. Bioorg Med Chem Lett20:1368-72 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calcitonin gene-related peptide type 1 receptor |
|---|
| Name: | Calcitonin gene-related peptide type 1 receptor |
|---|
| Synonyms: | Adrenomedullin receptor AM1; CALCRL/RAMP2 | CALCRL | CALRL_HUMAN | CGRP type 1 receptor | CGRP type 1 receptor mRNA | CGRPR | Calcitonin gene-related peptide (CGRP) receptor | Calcitonin gene-related peptide 1 (CGRP) | Calcitonin receptor-like receptor | Calcitonin receptor-like receptor (CLR) | Human CL receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 52980.45 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16602 |
|---|
| Residue: | 461 |
|---|
| Sequence: | MEKKCTLYFLVLLPFFMILVTAELEESPEDSIQLGVTRNKIMTAQYECYQKIMQDPIQQA
EGVYCNRTWDGWLCWNDVAAGTESMQLCPDYFQDFDPSEKVTKICDQDGNWFRHPASNRT
WTNYTQCNVNTHEKVKTALNLFYLTIIGHGLSIASLLISLGIFFYFKSLSCQRITLHKNL
FFSFVCNSVVTIIHLTAVANNQALVATNPVSCKVSQFIHLYLMGCNYFWMLCEGIYLHTL
IVVAVFAEKQHLMWYYFLGWGFPLIPACIHAIARSLYYNDNCWISSDTHLLYIIHGPICA
ALLVNLFFLLNIVRVLITKLKVTHQAESNLYMKAVRATLILVPLLGIEFVLIPWRPEGKI
AEEVYDYIMHILMHFQGLLVSTIFCFFNGEVQAILRRNWNQYKIQFGNSFSNSEALRSAS
YTVSTISDGPGYSHDCPSEHLNGKSIHDIENVLLKPENLYN
|
|
|
|---|
| BDBM50415360 |
|---|
| n/a |
|---|
| Name | BDBM50415360 |
|---|
| Synonyms: | CHEMBL610062 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H32ClN7O3 |
|---|
| Mol. Mass. | 550.052 |
|---|
| SMILES | CCN1CCC(CC1)c1cc(Cl)cc(c1)-c1nnc(CC(=O)N2CCC(CC2)n2c3cccnc3[nH]c2=O)o1 |
|---|
| Structure | 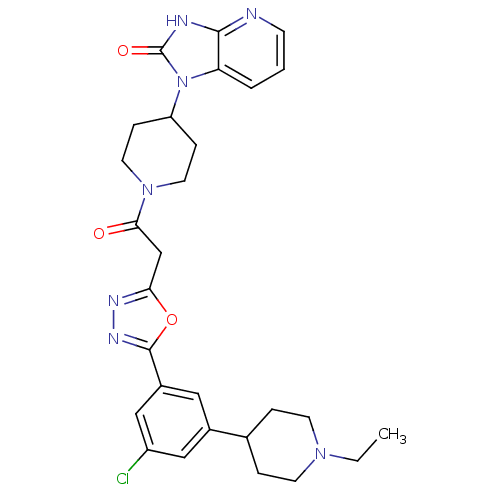
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nichols, PL; Brand, J; Briggs, M; D'Angeli, M; Farge, J; Garland, SL; Goldsmith, P; Hutchings, R; Kilford, I; Li, HY; MacPherson, D; Nimmo, F; Sanderson, FD; Sehmi, S; Shuker, N; Skidmore, J; Stott, M; Sweeting, J; Tajuddin, H; Takle, AK; Trani, G; Wall, ID; Ward, R; Wilson, DM; Witty, D Potent oxadiazole CGRP receptor antagonists for the potential treatment of migraine. Bioorg Med Chem Lett20:1368-72 (2010) [PubMed] Article
Nichols, PL; Brand, J; Briggs, M; D'Angeli, M; Farge, J; Garland, SL; Goldsmith, P; Hutchings, R; Kilford, I; Li, HY; MacPherson, D; Nimmo, F; Sanderson, FD; Sehmi, S; Shuker, N; Skidmore, J; Stott, M; Sweeting, J; Tajuddin, H; Takle, AK; Trani, G; Wall, ID; Ward, R; Wilson, DM; Witty, D Potent oxadiazole CGRP receptor antagonists for the potential treatment of migraine. Bioorg Med Chem Lett20:1368-72 (2010) [PubMed] Article