| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 8 |
|---|
| Ligand | BDBM50172920 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_621738 (CHEMBL1108470) |
|---|
| IC50 | 79.43±n/a nM |
|---|
| Citation |  Christopher, JA; Atkinson, FL; Bax, BD; Brown, MJ; Champigny, AC; Chuang, TT; Jones, EJ; Mosley, JE; Musgrave, JR 1-Aryl-3,4-dihydroisoquinoline inhibitors of JNK3. Bioorg Med Chem Lett19:2230-4 (2009) [PubMed] Article Christopher, JA; Atkinson, FL; Bax, BD; Brown, MJ; Champigny, AC; Chuang, TT; Jones, EJ; Mosley, JE; Musgrave, JR 1-Aryl-3,4-dihydroisoquinoline inhibitors of JNK3. Bioorg Med Chem Lett19:2230-4 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 8 |
|---|
| Name: | Mitogen-activated protein kinase 8 |
|---|
| Synonyms: | JNK-46 | JNK1 | JNK1-alpha-1 | MAPK8 | MK08_HUMAN | Mitogen-Activated Protein Kinase 8 (JNK1) | PRKM8 | SAPK1 | SAPK1C | Stress-activated protein kinase JNK1 | c-Jun N-terminal kinase 1 | c-Jun N-terminal kinase 1 (JNK1) | c-Jun N-terminal kinase 1(JNK1) | c-Jun N-terminal kinase 2 (JNK2) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48297.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | JNK-1 was purchased from Upstate Cell Signaling Solutions (formerly Upstate Biotechnology). |
|---|
| Residue: | 427 |
|---|
| Sequence: | MSRSKRDNNFYSVEIGDSTFTVLKRYQNLKPIGSGAQGIVCAAYDAILERNVAIKKLSRP
FQNQTHAKRAYRELVLMKCVNHKNIIGLLNVFTPQKSLEEFQDVYIVMELMDANLCQVIQ
MELDHERMSYLLYQMLCGIKHLHSAGIIHRDLKPSNIVVKSDCTLKILDFGLARTAGTSF
MMTPYVVTRYYRAPEVILGMGYKENVDLWSVGCIMGEMVCHKILFPGRDYIDQWNKVIEQ
LGTPCPEFMKKLQPTVRTYVENRPKYAGYSFEKLFPDVLFPADSEHNKLKASQARDLLSK
MLVIDASKRISVDEALQHPYINVWYDPSEAEAPPPKIPDKQLDEREHTIEEWKELIYKEV
MDLEERTKNGVIRGQPSPLGAAVINGSQHPSSSSSVNDVSSMSTDPTLASDTDSSLEAAA
GPLGCCR
|
|
|
|---|
| BDBM50172920 |
|---|
| n/a |
|---|
| Name | BDBM50172920 |
|---|
| Synonyms: | 3-(6-(2-chlorophenylamino)-1H-indazol-3-yl)-N-(piperidin-4-yl)benzamide | 3-[6-(2-Chloro-phenylamino)-1H-indazol-3-yl]-N-piperidin-4-yl-benzamide | CHEMBL194806 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H24ClN5O |
|---|
| Mol. Mass. | 445.944 |
|---|
| SMILES | Clc1ccccc1Nc1ccc2c(n[nH]c2c1)-c1cccc(c1)C(=O)NC1CCNCC1 |
|---|
| Structure | 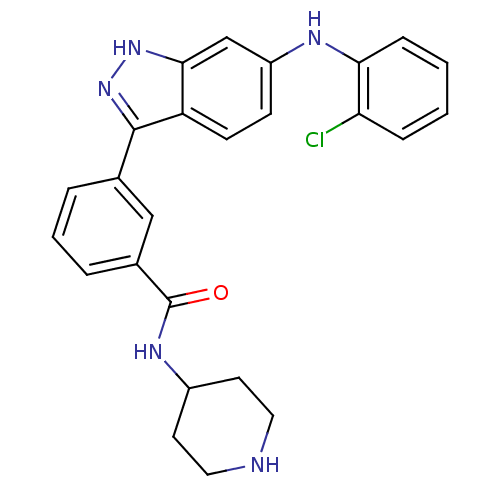
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Christopher, JA; Atkinson, FL; Bax, BD; Brown, MJ; Champigny, AC; Chuang, TT; Jones, EJ; Mosley, JE; Musgrave, JR 1-Aryl-3,4-dihydroisoquinoline inhibitors of JNK3. Bioorg Med Chem Lett19:2230-4 (2009) [PubMed] Article
Christopher, JA; Atkinson, FL; Bax, BD; Brown, MJ; Champigny, AC; Chuang, TT; Jones, EJ; Mosley, JE; Musgrave, JR 1-Aryl-3,4-dihydroisoquinoline inhibitors of JNK3. Bioorg Med Chem Lett19:2230-4 (2009) [PubMed] Article