| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H4 receptor |
|---|
| Ligand | BDBM50418061 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_735234 (CHEMBL1694496) |
|---|
| Ki | 89.13±n/a nM |
|---|
| Citation |  Wijtmans, M; de Graaf, C; de Kloe, G; Istyastono, EP; Smit, J; Lim, H; Boonnak, R; Nijmeijer, S; Smits, RA; Jongejan, A; Zuiderveld, O; de Esch, IJ; Leurs, R Triazole ligands reveal distinct molecular features that induce histamine H4 receptor affinity and subtly govern H4/H3 subtype selectivity. J Med Chem54:1693-703 (2011) [PubMed] Article Wijtmans, M; de Graaf, C; de Kloe, G; Istyastono, EP; Smit, J; Lim, H; Boonnak, R; Nijmeijer, S; Smits, RA; Jongejan, A; Zuiderveld, O; de Esch, IJ; Leurs, R Triazole ligands reveal distinct molecular features that induce histamine H4 receptor affinity and subtly govern H4/H3 subtype selectivity. J Med Chem54:1693-703 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H4 receptor |
|---|
| Name: | Histamine H4 receptor |
|---|
| Synonyms: | AXOR35 | G-protein coupled receptor 105 | GPCR105 | GPRv53 | HH4R | HISTAMINE H4 | HRH4 | HRH4_HUMAN | Histamine H4 receptor | Histamine H4 receptor (H4R) | Histamine receptor (H3 and H4) | Pfi-013 | SP9144 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44517.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH4R receptors. |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPDTNSTINLSLSTRVTLAFFMSLVAFAIMLGNALVILAFVVDKNLRHRSSYFFLNLAIS
DFFVGVISIPLYIPHTLFEWDFGKEICVFWLTTDYLLCTASVYNIVLISYDRYLSVSNAV
SYRTQHTGVLKIVTLMVAVWVLAFLVNGPMILVSESWKDEGSECEPGFFSEWYILAITSF
LEFVIPVILVAYFNMNIYWSLWKRDHLSRCQSHPGLTAVSSNICGHSFRGRLSSRRSLSA
STEVPASFHSERQRRKSSLMFSSRTKMNSNTIASKMGSFSQSDSVALHQREHVELLRARR
LAKSLAILLGVFAVCWAPYSLFTIVLSFYSSATGPKSVWYRIAFWLQWFNSFVNPLLYPL
CHKRFQKAFLKIFCIKKQPLPSQHSRSVSS
|
|
|
|---|
| BDBM50418061 |
|---|
| n/a |
|---|
| Name | BDBM50418061 |
|---|
| Synonyms: | CHEMBL1688971 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H21N5 |
|---|
| Mol. Mass. | 259.35 |
|---|
| SMILES | C(Cc1cnc[nH]1)Cn1cc(nn1)C1CCCCC1 |
|---|
| Structure | 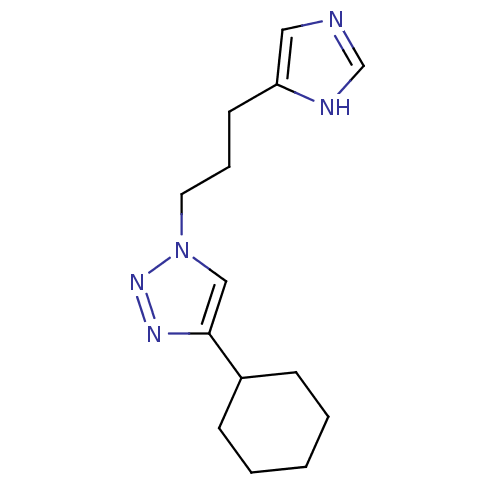
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wijtmans, M; de Graaf, C; de Kloe, G; Istyastono, EP; Smit, J; Lim, H; Boonnak, R; Nijmeijer, S; Smits, RA; Jongejan, A; Zuiderveld, O; de Esch, IJ; Leurs, R Triazole ligands reveal distinct molecular features that induce histamine H4 receptor affinity and subtly govern H4/H3 subtype selectivity. J Med Chem54:1693-703 (2011) [PubMed] Article
Wijtmans, M; de Graaf, C; de Kloe, G; Istyastono, EP; Smit, J; Lim, H; Boonnak, R; Nijmeijer, S; Smits, RA; Jongejan, A; Zuiderveld, O; de Esch, IJ; Leurs, R Triazole ligands reveal distinct molecular features that induce histamine H4 receptor affinity and subtly govern H4/H3 subtype selectivity. J Med Chem54:1693-703 (2011) [PubMed] Article