| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM28661 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_741372 (CHEMBL1764898) |
|---|
| IC50 | 1995±n/a nM |
|---|
| Citation |  Evans, KA; Shearer, BG; Wisnoski, DD; Shi, D; Sparks, SM; Sternbach, DD; Winegar, DA; Billin, AN; Britt, C; Way, JM; Epperly, AH; Leesnitzer, LM; Merrihew, RV; Xu, RX; Lambert, MH; Jin, J Phenoxyacetic acids as PPARd partial agonists: synthesis, optimization, and in vivo efficacy. Bioorg Med Chem Lett21:2345-50 (2011) [PubMed] Article Evans, KA; Shearer, BG; Wisnoski, DD; Shi, D; Sparks, SM; Sternbach, DD; Winegar, DA; Billin, AN; Britt, C; Way, JM; Epperly, AH; Leesnitzer, LM; Merrihew, RV; Xu, RX; Lambert, MH; Jin, J Phenoxyacetic acids as PPARd partial agonists: synthesis, optimization, and in vivo efficacy. Bioorg Med Chem Lett21:2345-50 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52222.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07869 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSC
PGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFV
IHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDI
FLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM28661 |
|---|
| n/a |
|---|
| Name | BDBM28661 |
|---|
| Synonyms: | 2-{2-methyl-4-[({4-methyl-2-[4-(trifluoromethyl)phenyl]-1,3-thiazol-5-yl}methyl)sulfanyl]phenoxy}acetic acid | CHEMBL38943 | GW 501516 | GW-501516 | GW1516 | GW501516 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H18F3NO3S2 |
|---|
| Mol. Mass. | 453.498 |
|---|
| SMILES | Cc1nc(sc1CSc1ccc(OCC(O)=O)c(C)c1)-c1ccc(cc1)C(F)(F)F |
|---|
| Structure | 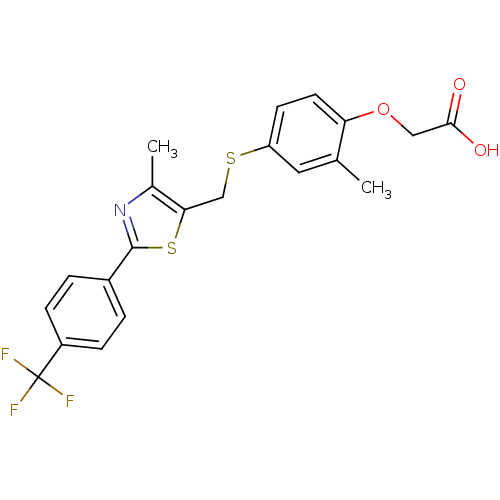
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Evans, KA; Shearer, BG; Wisnoski, DD; Shi, D; Sparks, SM; Sternbach, DD; Winegar, DA; Billin, AN; Britt, C; Way, JM; Epperly, AH; Leesnitzer, LM; Merrihew, RV; Xu, RX; Lambert, MH; Jin, J Phenoxyacetic acids as PPARd partial agonists: synthesis, optimization, and in vivo efficacy. Bioorg Med Chem Lett21:2345-50 (2011) [PubMed] Article
Evans, KA; Shearer, BG; Wisnoski, DD; Shi, D; Sparks, SM; Sternbach, DD; Winegar, DA; Billin, AN; Britt, C; Way, JM; Epperly, AH; Leesnitzer, LM; Merrihew, RV; Xu, RX; Lambert, MH; Jin, J Phenoxyacetic acids as PPARd partial agonists: synthesis, optimization, and in vivo efficacy. Bioorg Med Chem Lett21:2345-50 (2011) [PubMed] Article