

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 1A2 | ||
| Ligand | BDBM50419562 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_796518 (CHEMBL1937833) | ||
| IC50 | <10000±n/a nM | ||
| Citation |  Morley, AD; King, S; Roberts, B; Lever, S; Teobald, B; Fisher, A; Cook, T; Parker, B; Wenlock, M; Phillips, C; Grime, K Lead optimisation of pyrazoles as novel FPR1 antagonists. Bioorg Med Chem Lett22:532-6 (2011) [PubMed] Article Morley, AD; King, S; Roberts, B; Lever, S; Teobald, B; Fisher, A; Cook, T; Parker, B; Wenlock, M; Phillips, C; Grime, K Lead optimisation of pyrazoles as novel FPR1 antagonists. Bioorg Med Chem Lett22:532-6 (2011) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 1A2 | |||
| Name: | Cytochrome P450 1A2 | ||
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 58423.38 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P05177 | ||
| Residue: | 516 | ||
| Sequence: |
| ||
| BDBM50419562 | |||
| n/a | |||
| Name | BDBM50419562 | ||
| Synonyms: | CHEMBL1934424 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C25H38N4O4S | ||
| Mol. Mass. | 490.659 | ||
| SMILES | CO[C@H]1CC[C@H](CC1)n1nc(C)c(C(=O)N[C@@H](C)C(C)(C)C)c1NS(=O)(=O)c1ccc(C)cc1 |r,wU:5.8,16.17,2.1,(31.27,-8.16,;32.01,-6.81,;31.21,-5.49,;29.67,-5.52,;28.87,-4.21,;29.61,-2.86,;31.15,-2.82,;31.95,-4.13,;29.13,-1.41,;27.66,-.94,;27.66,.6,;26.41,1.51,;29.12,1.08,;29.59,2.55,;31.1,2.88,;28.56,3.69,;29.03,5.16,;27.99,6.3,;30.53,5.48,;31.57,4.34,;31,6.95,;32.01,5.9,;30.03,-.15,;31.57,-.15,;32.33,1.2,;30.99,1.97,;32.33,2.74,;33.88,1.2,;34.64,-.13,;36.18,-.13,;36.95,1.2,;38.49,1.2,;36.18,2.53,;34.64,2.52,)| | ||
| Structure | 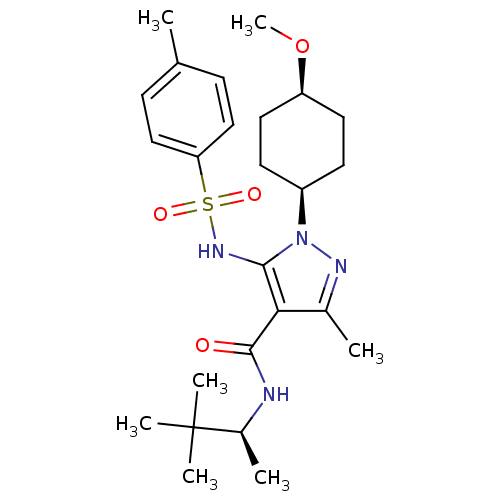
| ||