| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Kappa-type opioid receptor |
|---|
| Ligand | BDBM50083230 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_839811 (CHEMBL2089822) |
|---|
| Ki | >400±n/a nM |
|---|
| Citation |  Pedregal, C; Joshi, EM; Toledo, MA; Lafuente, C; Diaz, N; Martinez-Grau, MA; Jiménez, A; Benito, A; Navarro, A; Chen, Z; Mudra, DR; Kahl, SD; Rash, KS; Statnick, MA; Barth, VN Development of LC-MS/MS-based receptor occupancy tracers and positron emission tomography radioligands for the nociceptin/orphanin FQ (NOP) receptor. J Med Chem55:4955-67 (2012) [PubMed] Article Pedregal, C; Joshi, EM; Toledo, MA; Lafuente, C; Diaz, N; Martinez-Grau, MA; Jiménez, A; Benito, A; Navarro, A; Chen, Z; Mudra, DR; Kahl, SD; Rash, KS; Statnick, MA; Barth, VN Development of LC-MS/MS-based receptor occupancy tracers and positron emission tomography radioligands for the nociceptin/orphanin FQ (NOP) receptor. J Med Chem55:4955-67 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Kappa-type opioid receptor |
|---|
| Name: | Kappa-type opioid receptor |
|---|
| Synonyms: | K-OR-1 | KOR-1 | Kappa-opioid receptor (KOR) | Kappa-type opioid receptor (KOPR) | Kappa-type opioid receptor (KOR) | Kappa-type opioid receptor (Kappa) | OPIATE Kappa | OPRK | OPRK1 | OPRK_HUMAN | kappa opioid receptor (KOR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 42648.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P41145 |
|---|
| Residue: | 380 |
|---|
| Sequence: | MDSPIQIFRGEPGPTCAPSACLPPNSSAWFPGWAEPDSNGSAGSEDAQLEPAHISPAIPV
IITAVYSVVFVVGLVGNSLVMFVIIRYTKMKTATNIYIFNLALADALVTTTMPFQSTVYL
MNSWPFGDVLCKIVISIDYYNMFTSIFTLTMMSVDRYIAVCHPVKALDFRTPLKAKIINI
CIWLLSSSVGISAIVLGGTKVREDVDVIECSLQFPDDDYSWWDLFMKICVFIFAFVIPVL
IIIVCYTLMILRLKSVRLLSGSREKDRNLRRITRLVLVVVAVFVVCWTPIHIFILVEALG
STSHSTAALSSYYFCIALGYTNSSLNPILYAFLDENFKRCFRDFCFPLKMRMERQSTSRV
RNTVQDPAYLRDIDGMNKPV
|
|
|
|---|
| BDBM50083230 |
|---|
| n/a |
|---|
| Name | BDBM50083230 |
|---|
| Synonyms: | 1-((3R,4R)-1-Cyclooctylmethyl-3-hydroxymethyl-piperidin-4-yl)-3-ethyl-1,3-dihydro-benzoimidazol-2-one | 1-[(3R,4R)-1-cyclooctylmethyl-3-hydroxymethyl-4-piperidinyl]-3-ethyl-1,3-dihydro-2H-benzimidazol-2-one | 1-[(3R,4R)-1-cyclooctylmethyl-3-hydroxymethyl-4-piperidyl]-3-ethyl-1,3-dihydro-2H-benzimidazol-2-one | CHEMBL357076 | J-113397 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H37N3O2 |
|---|
| Mol. Mass. | 399.5695 |
|---|
| SMILES | CCn1c2ccccc2n([C@@H]2CCN(CC3CCCCCCC3)C[C@H]2CO)c1=O |
|---|
| Structure | 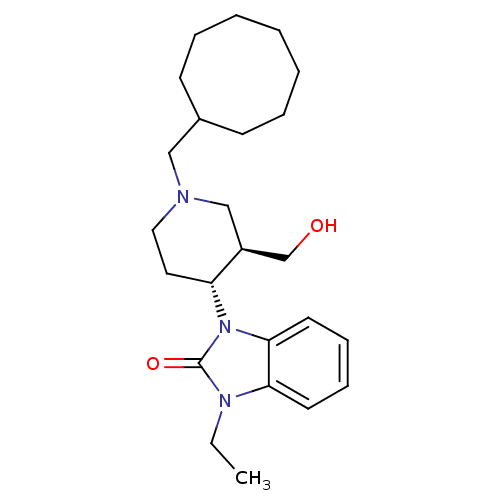
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pedregal, C; Joshi, EM; Toledo, MA; Lafuente, C; Diaz, N; Martinez-Grau, MA; Jiménez, A; Benito, A; Navarro, A; Chen, Z; Mudra, DR; Kahl, SD; Rash, KS; Statnick, MA; Barth, VN Development of LC-MS/MS-based receptor occupancy tracers and positron emission tomography radioligands for the nociceptin/orphanin FQ (NOP) receptor. J Med Chem55:4955-67 (2012) [PubMed] Article
Pedregal, C; Joshi, EM; Toledo, MA; Lafuente, C; Diaz, N; Martinez-Grau, MA; Jiménez, A; Benito, A; Navarro, A; Chen, Z; Mudra, DR; Kahl, SD; Rash, KS; Statnick, MA; Barth, VN Development of LC-MS/MS-based receptor occupancy tracers and positron emission tomography radioligands for the nociceptin/orphanin FQ (NOP) receptor. J Med Chem55:4955-67 (2012) [PubMed] Article