| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50067482 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_41557 (CHEMBL857083) |
|---|
| IC50 | 11749±n/a nM |
|---|
| Citation |  Bolognesi, ML; Andrisano, V; Bartolini, M; Minarini, A; Rosini, M; Tumiatti, V; Melchiorre, C Hexahydrochromeno[4,3-b]pyrrole derivatives as acetylcholinesterase inhibitors. J Med Chem44:105-9 (2001) [PubMed] Bolognesi, ML; Andrisano, V; Bartolini, M; Minarini, A; Rosini, M; Tumiatti, V; Melchiorre, C Hexahydrochromeno[4,3-b]pyrrole derivatives as acetylcholinesterase inhibitors. J Med Chem44:105-9 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | Acylcholine acylhydrolase | BCHE | Butyrylcholine esterase (BChE) | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | CHE1 | CHLE_HUMAN | Choline esterase II | Cholinesterases | Cholinesterases; ACHE & BCHE | Pseudocholinesterase |
|---|
| Type: | Homotetramer |
|---|
| Mol. Mass.: | 68422.27 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06276 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MHSKVTIICIRFLFWFLLLCMLIGKSHTEDDIIIATKNGKVRGMNLTVFGGTVTAFLGIP
YAQPPLGRLRFKKPQSLTKWSDIWNATKYANSCCQNIDQSFPGFHGSEMWNPNTDLSEDC
LYLNVWIPAPKPKNATVLIWIYGGGFQTGTSSLHVYDGKFLARVERVIVVSMNYRVGALG
FLALPGNPEAPGNMGLFDQQLALQWVQKNIAAFGGNPKSVTLFGESAGAASVSLHLLSPG
SHSLFTRAILQSGSFNAPWAVTSLYEARNRTLNLAKLTGCSRENETEIIKCLRNKDPQEI
LLNEAFVVPYGTPLSVNFGPTVDGDFLTDMPDILLELGQFKKTQILVGVNKDEGTAFLVY
GAPGFSKDNNSIITRKEFQEGLKIFFPGVSEFGKESILFHYTDWVDDQRPENYREALGDV
VGDYNFICPALEFTKKFSEWGNNAFFYYFEHRSSKLPWPEWMGVMHGYEIEFVFGLPLER
RDNYTKAEEILSRSIVKRWANFAKYGNPNETQNNSTSWPVFKSTEQKYLTLNTESTRIMT
KLRAQQCRFWTSFFPKVLEMTGNIDEAEWEWKAGFHRWNNYMMDWKNQFNDYTSKKESCV
GL
|
|
|
|---|
| BDBM50067482 |
|---|
| n/a |
|---|
| Name | BDBM50067482 |
|---|
| Synonyms: | 6-[(2-Methoxy-benzyl)-methyl-amino]-hexanoic acid [8-({6-[(2-methoxy-benzyl)-methyl-amino]-hexanoyl}-methyl-amino)-octyl]-methyl-amide | CAPROCTAMINE | CHEMBL11805 | CHEMBL222856 | CHEMBL557586 | N,N''-(octane-1,8-diyl)bis(6-((2-methoxybenzyl)(methyl)amino)-N-methylhexanamide) |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C40H66N4O4 |
|---|
| Mol. Mass. | 666.9764 |
|---|
| SMILES | COc1ccccc1CN(C)CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(C)Cc1ccccc1OC |
|---|
| Structure | 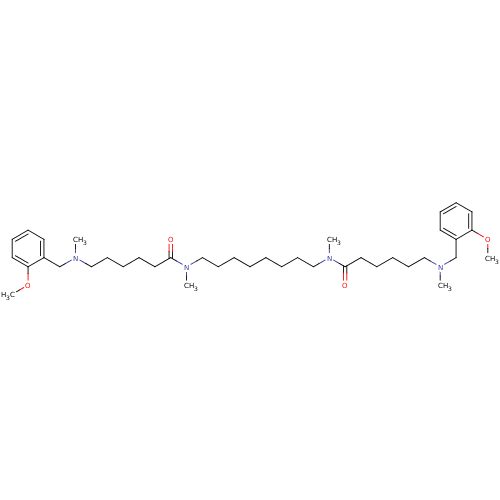
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bolognesi, ML; Andrisano, V; Bartolini, M; Minarini, A; Rosini, M; Tumiatti, V; Melchiorre, C Hexahydrochromeno[4,3-b]pyrrole derivatives as acetylcholinesterase inhibitors. J Med Chem44:105-9 (2001) [PubMed]
Bolognesi, ML; Andrisano, V; Bartolini, M; Minarini, A; Rosini, M; Tumiatti, V; Melchiorre, C Hexahydrochromeno[4,3-b]pyrrole derivatives as acetylcholinesterase inhibitors. J Med Chem44:105-9 (2001) [PubMed]