| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase ZAP-70 |
|---|
| Ligand | BDBM50432373 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_952222 (CHEMBL2351281) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Kawakita, Y; Seto, M; Ohashi, T; Tamura, T; Yusa, T; Miki, H; Iwata, H; Kamiguchi, H; Tanaka, T; Sogabe, S; Ohta, Y; Ishikawa, T Design and synthesis of novel pyrimido[4,5-b]azepine derivatives as HER2/EGFR dual inhibitors. Bioorg Med Chem21:2250-61 (2013) [PubMed] Article Kawakita, Y; Seto, M; Ohashi, T; Tamura, T; Yusa, T; Miki, H; Iwata, H; Kamiguchi, H; Tanaka, T; Sogabe, S; Ohta, Y; Ishikawa, T Design and synthesis of novel pyrimido[4,5-b]azepine derivatives as HER2/EGFR dual inhibitors. Bioorg Med Chem21:2250-61 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase ZAP-70 |
|---|
| Name: | Tyrosine-protein kinase ZAP-70 |
|---|
| Synonyms: | 70 kDa zeta-associated protein | SRK | Syk-related tyrosine kinase | Tyrosine Kinase ZAP-70 | Tyrosine-protein kinase ZAP-70 | Tyrosine-protein kinase ZAP-70 (Syk) | Tyrosine-protein kinase ZAP-70 (ZAP70) | Tyrosine-protein kinase ZAP70 | ZAP70 | ZAP70_HUMAN | Zeta-chain (TCR) associated protein kinase 70kDa |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 69881.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ZAP-70 SH2 domain was expressed and purified from E. coli using O-phospho-L-tyrosine-agarose column. |
|---|
| Residue: | 619 |
|---|
| Sequence: | MPDPAAHLPFFYGSISRAEAEEHLKLAGMADGLFLLRQCLRSLGGYVLSLVHDVRFHHFP
IERQLNGTYAIAGGKAHCGPAELCEFYSRDPDGLPCNLRKPCNRPSGLEPQPGVFDCLRD
AMVRDYVRQTWKLEGEALEQAIISQAPQVEKLIATTAHERMPWYHSSLTREEAERKLYSG
AQTDGKFLLRPRKEQGTYALSLIYGKTVYHYLISQDKAGKYCIPEGTKFDTLWQLVEYLK
LKADGLIYCLKEACPNSSASNASGAAAPTLPAHPSTLTHPQRRIDTLNSDGYTPEPARIT
SPDKPRPMPMDTSVYESPYSDPEELKDKKLFLKRDNLLIADIELGCGNFGSVRQGVYRMR
KKQIDVAIKVLKQGTEKADTEEMMREAQIMHQLDNPYIVRLIGVCQAEALMLVMEMAGGG
PLHKFLVGKREEIPVSNVAELLHQVSMGMKYLEEKNFVHRDLAARNVLLVNRHYAKISDF
GLSKALGADDSYYTARSAGKWPLKWYAPECINFRKFSSRSDVWSYGVTMWEALSYGQKPY
KKMKGPEVMAFIEQGKRMECPPECPPELYALMSDCWIYKWEDRPDFLTVEQRMRACYYSL
ASKVEGPPGSTQKAEAACA
|
|
|
|---|
| BDBM50432373 |
|---|
| n/a |
|---|
| Name | BDBM50432373 |
|---|
| Synonyms: | CHEMBL2348417 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H22ClN5O3S |
|---|
| Mol. Mass. | 507.992 |
|---|
| SMILES | OCCNC(=O)C1=Cc2c(Nc3ccc(Oc4cccc5sccc45)c(Cl)c3)ncnc2NCC1 |t:6| |
|---|
| Structure | 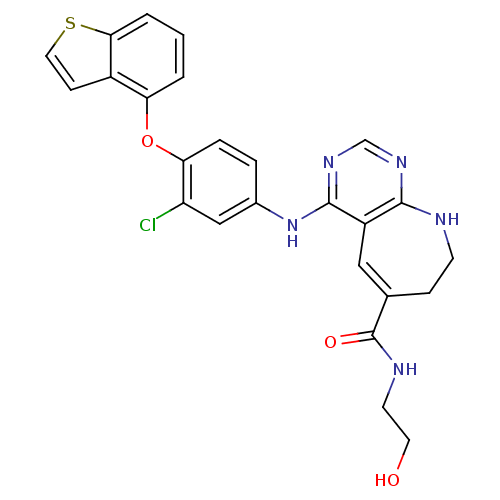
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kawakita, Y; Seto, M; Ohashi, T; Tamura, T; Yusa, T; Miki, H; Iwata, H; Kamiguchi, H; Tanaka, T; Sogabe, S; Ohta, Y; Ishikawa, T Design and synthesis of novel pyrimido[4,5-b]azepine derivatives as HER2/EGFR dual inhibitors. Bioorg Med Chem21:2250-61 (2013) [PubMed] Article
Kawakita, Y; Seto, M; Ohashi, T; Tamura, T; Yusa, T; Miki, H; Iwata, H; Kamiguchi, H; Tanaka, T; Sogabe, S; Ohta, Y; Ishikawa, T Design and synthesis of novel pyrimido[4,5-b]azepine derivatives as HER2/EGFR dual inhibitors. Bioorg Med Chem21:2250-61 (2013) [PubMed] Article