| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase JAK3 |
|---|
| Ligand | BDBM50434787 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_962473 (CHEMBL2390366) |
|---|
| Ki | 5.9±n/a nM |
|---|
| Citation |  Zak, M; Hurley, CA; Ward, SI; Bergeron, P; Barrett, K; Balazs, M; Blair, WS; Bull, R; Chakravarty, P; Chang, C; Crackett, P; Deshmukh, G; DeVoss, J; Dragovich, PS; Eigenbrot, C; Ellwood, C; Gaines, S; Ghilardi, N; Gibbons, P; Gradl, S; Gribling, P; Hamman, C; Harstad, E; Hewitt, P; Johnson, A; Johnson, T; Kenny, JR; Koehler, MF; Bir Kohli, P; Labadie, S; Lee, WP; Liao, J; Liimatta, M; Mendonca, R; Narukulla, R; Pulk, R; Reeve, A; Savage, S; Shia, S; Steffek, M; Ubhayakar, S; van Abbema, A; Aliagas, I; Avitabile-Woo, B; Xiao, Y; Yang, J; Kulagowski, JJ Identification of C-2 hydroxyethyl imidazopyrrolopyridines as potent JAK1 inhibitors with favorable physicochemical properties and high selectivity over JAK2. J Med Chem56:4764-85 (2013) [PubMed] Article Zak, M; Hurley, CA; Ward, SI; Bergeron, P; Barrett, K; Balazs, M; Blair, WS; Bull, R; Chakravarty, P; Chang, C; Crackett, P; Deshmukh, G; DeVoss, J; Dragovich, PS; Eigenbrot, C; Ellwood, C; Gaines, S; Ghilardi, N; Gibbons, P; Gradl, S; Gribling, P; Hamman, C; Harstad, E; Hewitt, P; Johnson, A; Johnson, T; Kenny, JR; Koehler, MF; Bir Kohli, P; Labadie, S; Lee, WP; Liao, J; Liimatta, M; Mendonca, R; Narukulla, R; Pulk, R; Reeve, A; Savage, S; Shia, S; Steffek, M; Ubhayakar, S; van Abbema, A; Aliagas, I; Avitabile-Woo, B; Xiao, Y; Yang, J; Kulagowski, JJ Identification of C-2 hydroxyethyl imidazopyrrolopyridines as potent JAK1 inhibitors with favorable physicochemical properties and high selectivity over JAK2. J Med Chem56:4764-85 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase JAK3 |
|---|
| Name: | Tyrosine-protein kinase JAK3 |
|---|
| Synonyms: | JAK-3 | JAK3 | JAK3_HUMAN | Janus kinase 3 | Janus kinase 3 (JAK3) | Janus kinase 3 JAK3 | L-JAK | Leukocyte janus kinase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 125111.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P52333 |
|---|
| Residue: | 1124 |
|---|
| Sequence: | MAPPSEETPLIPQRSCSLLSTEAGALHVLLPARGPGPPQRLSFSFGDHLAEDLCVQAAKA
SGILPVYHSLFALATEDLSCWFPPSHIFSVEDASTQVLLYRIRFYFPNWFGLEKCHRFGL
RKDLASAILDLPVLEHLFAQHRSDLVSGRLPVGLSLKEQGECLSLAVLDLARMAREQAQR
PGELLKTVSYKACLPPSLRDLIQGLSFVTRRRIRRTVRRALRRVAACQADRHSLMAKYIM
DLERLDPAGAAETFHVGLPGALGGHDGLGLLRVAGDGGIAWTQGEQEVLQPFCDFPEIVD
ISIKQAPRVGPAGEHRLVTVTRTDNQILEAEFPGLPEALSFVALVDGYFRLTTDSQHFFC
KEVAPPRLLEEVAEQCHGPITLDFAINKLKTGGSRPGSYVLRRSPQDFDSFLLTVCVQNP
LGPDYKGCLIRRSPTGTFLLVGLSRPHSSLRELLATCWDGGLHVDGVAVTLTSCCIPRPK
EKSNLIVVQRGHSPPTSSLVQPQSQYQLSQMTFHKIPADSLEWHENLGHGSFTKIYRGCR
HEVVDGEARKTEVLLKVMDAKHKNCMESFLEAASLMSQVSYRHLVLLHGVCMAGDSTMVQ
EFVHLGAIDMYLRKRGHLVPASWKLQVVKQLAYALNYLEDKGLPHGNVSARKVLLAREGA
DGSPPFIKLSDPGVSPAVLSLEMLTDRIPWVAPECLREAQTLSLEADKWGFGATVWEVFS
GVTMPISALDPAKKLQFYEDRQQLPAPKWTELALLIQQCMAYEPVQRPSFRAVIRDLNSL
ISSDYELLSDPTPGALAPRDGLWNGAQLYACQDPTIFEERHLKYISQLGKGNFGSVELCR
YDPLGDNTGALVAVKQLQHSGPDQQRDFQREIQILKALHSDFIVKYRGVSYGPGRQSLRL
VMEYLPSGCLRDFLQRHRARLDASRLLLYSSQICKGMEYLGSRRCVHRDLAARNILVESE
AHVKIADFGLAKLLPLDKDYYVVREPGQSPIFWYAPESLSDNIFSRQSDVWSFGVVLYEL
FTYCDKSCSPSAEFLRMMGCERDVPALCRLLELLEEGQRLPAPPACPAEVHELMKLCWAP
SPQDRPSFSALGPQLDMLWSGSRGCETHAFTAHPEGKHHSLSFS
|
|
|
|---|
| BDBM50434787 |
|---|
| n/a |
|---|
| Name | BDBM50434787 |
|---|
| Synonyms: | CHEMBL2386635 | US10487083, Example C | US10703751, Example 644 of WO2011086053 | US11059823, Example C |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H21N5O |
|---|
| Mol. Mass. | 323.3922 |
|---|
| SMILES | C[C@@H](O)c1nc2cnc3[nH]ccc3c2n1[C@H]1CC[C@H](CC#N)CC1 |r,wU:18.21,wD:15.17,1.1,(49.07,-5.95,;48.31,-7.29,;46.77,-7.3,;49.09,-8.62,;48.48,-10.02,;49.62,-11.04,;49.62,-12.58,;50.95,-13.35,;52.28,-12.58,;53.75,-13.05,;54.65,-11.81,;53.75,-10.56,;52.28,-11.04,;50.94,-10.26,;50.61,-8.77,;51.68,-7.66,;53.17,-8.05,;54.24,-6.95,;53.83,-5.47,;54.9,-4.37,;56.39,-4.75,;57.88,-5.12,;52.34,-5.09,;51.26,-6.19,)| |
|---|
| Structure | 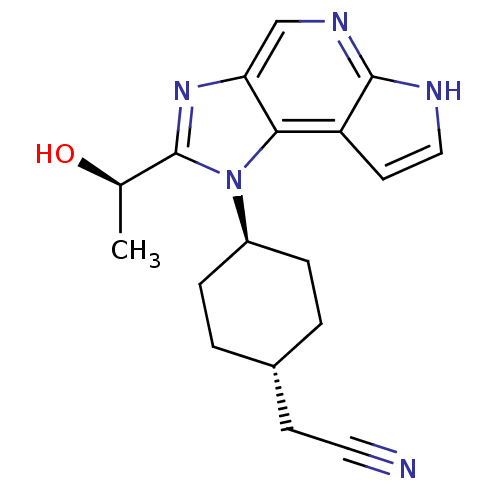
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zak, M; Hurley, CA; Ward, SI; Bergeron, P; Barrett, K; Balazs, M; Blair, WS; Bull, R; Chakravarty, P; Chang, C; Crackett, P; Deshmukh, G; DeVoss, J; Dragovich, PS; Eigenbrot, C; Ellwood, C; Gaines, S; Ghilardi, N; Gibbons, P; Gradl, S; Gribling, P; Hamman, C; Harstad, E; Hewitt, P; Johnson, A; Johnson, T; Kenny, JR; Koehler, MF; Bir Kohli, P; Labadie, S; Lee, WP; Liao, J; Liimatta, M; Mendonca, R; Narukulla, R; Pulk, R; Reeve, A; Savage, S; Shia, S; Steffek, M; Ubhayakar, S; van Abbema, A; Aliagas, I; Avitabile-Woo, B; Xiao, Y; Yang, J; Kulagowski, JJ Identification of C-2 hydroxyethyl imidazopyrrolopyridines as potent JAK1 inhibitors with favorable physicochemical properties and high selectivity over JAK2. J Med Chem56:4764-85 (2013) [PubMed] Article
Zak, M; Hurley, CA; Ward, SI; Bergeron, P; Barrett, K; Balazs, M; Blair, WS; Bull, R; Chakravarty, P; Chang, C; Crackett, P; Deshmukh, G; DeVoss, J; Dragovich, PS; Eigenbrot, C; Ellwood, C; Gaines, S; Ghilardi, N; Gibbons, P; Gradl, S; Gribling, P; Hamman, C; Harstad, E; Hewitt, P; Johnson, A; Johnson, T; Kenny, JR; Koehler, MF; Bir Kohli, P; Labadie, S; Lee, WP; Liao, J; Liimatta, M; Mendonca, R; Narukulla, R; Pulk, R; Reeve, A; Savage, S; Shia, S; Steffek, M; Ubhayakar, S; van Abbema, A; Aliagas, I; Avitabile-Woo, B; Xiao, Y; Yang, J; Kulagowski, JJ Identification of C-2 hydroxyethyl imidazopyrrolopyridines as potent JAK1 inhibitors with favorable physicochemical properties and high selectivity over JAK2. J Med Chem56:4764-85 (2013) [PubMed] Article