| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Ileal sodium/bile acid cotransporter |
|---|
| Ligand | BDBM50134411 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_963355 (CHEMBL2388852) |
|---|
| IC50 | 22±n/a nM |
|---|
| Citation |  Wu, Y; Aquino, CJ; Cowan, DJ; Anderson, DL; Ambroso, JL; Bishop, MJ; Boros, EE; Chen, L; Cunningham, A; Dobbins, RL; Feldman, PL; Harston, LT; Kaldor, IW; Klein, R; Liang, X; McIntyre, MS; Merrill, CL; Patterson, KM; Prescott, JS; Ray, JS; Roller, SG; Yao, X; Young, A; Yuen, J; Collins, JL Discovery of a highly potent, nonabsorbable apical sodium-dependent bile acid transporter inhibitor (GSK2330672) for treatment of type 2 diabetes. J Med Chem56:5094-114 (2013) [PubMed] Article Wu, Y; Aquino, CJ; Cowan, DJ; Anderson, DL; Ambroso, JL; Bishop, MJ; Boros, EE; Chen, L; Cunningham, A; Dobbins, RL; Feldman, PL; Harston, LT; Kaldor, IW; Klein, R; Liang, X; McIntyre, MS; Merrill, CL; Patterson, KM; Prescott, JS; Ray, JS; Roller, SG; Yao, X; Young, A; Yuen, J; Collins, JL Discovery of a highly potent, nonabsorbable apical sodium-dependent bile acid transporter inhibitor (GSK2330672) for treatment of type 2 diabetes. J Med Chem56:5094-114 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Ileal sodium/bile acid cotransporter |
|---|
| Name: | Ileal sodium/bile acid cotransporter |
|---|
| Synonyms: | ASBT | Apical sodium-dependent bile acid transporter | IBAT | ISBT | Ileal Na(+)/bile acid cotransporter | Ileal sodium-dependent bile acid transporter | NTCP2_RAT | Na(+)-dependent ileal bile acid transporter | Ntcp2 | Slc10a2 | Sodium/taurocholate-cotransporting polypeptide, ileal | Solute carrier family 10 member 2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 38020.22 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_104627 |
|---|
| Residue: | 348 |
|---|
| Sequence: | MDNSSVCSPNATFCEGDSCLVTESNFNAILSTVMSTVLTILLAMVMFSMGCNVEINKFLG
HIKRPWGIFVGFLCQFGIMPLTGFILSVASGILPVQAVVVLIMGCCPGGTGSNILAYWID
GDMDLSVSMTTCSTLLALGMMPLCLFIYTKMWVDSGTIVIPYDSIGISLVALVIPVSIGM
FVNHKWPQKAKIILKIGSIAGAILIVLIAVVGGILYQSAWIIEPKLWIIGTIFPIAGYSL
GFFLARLAGQPWYRCRTVALETGMQNTQLCSTIVQLSFSPEDLNLVFTFPLIYTVFQLVF
AAIILGMYVTYKKCHGKNDAEFLEKTDNDMDPMPSFQETNKGFQPDEK
|
|
|
|---|
| BDBM50134411 |
|---|
| n/a |
|---|
| Name | BDBM50134411 |
|---|
| Synonyms: | (7R,9R)-7-Butyl-7-ethyl-2,3-dimethoxy-9-phenyl-6,7,8,9-tetrahydro-5-thia-8-aza-benzocycloheptene 5,5-dioxide | CHEMBL332370 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H31NO4S |
|---|
| Mol. Mass. | 417.562 |
|---|
| SMILES | CCCC[C@]1(CC)CS(=O)(=O)c2cc(OC)c(OC)cc2[C@H](N1)c1ccccc1 |
|---|
| Structure | 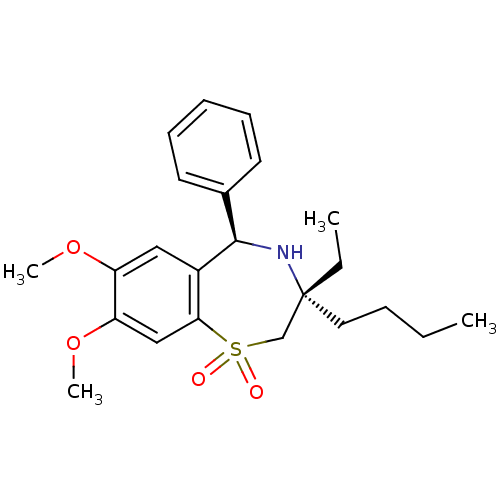
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wu, Y; Aquino, CJ; Cowan, DJ; Anderson, DL; Ambroso, JL; Bishop, MJ; Boros, EE; Chen, L; Cunningham, A; Dobbins, RL; Feldman, PL; Harston, LT; Kaldor, IW; Klein, R; Liang, X; McIntyre, MS; Merrill, CL; Patterson, KM; Prescott, JS; Ray, JS; Roller, SG; Yao, X; Young, A; Yuen, J; Collins, JL Discovery of a highly potent, nonabsorbable apical sodium-dependent bile acid transporter inhibitor (GSK2330672) for treatment of type 2 diabetes. J Med Chem56:5094-114 (2013) [PubMed] Article
Wu, Y; Aquino, CJ; Cowan, DJ; Anderson, DL; Ambroso, JL; Bishop, MJ; Boros, EE; Chen, L; Cunningham, A; Dobbins, RL; Feldman, PL; Harston, LT; Kaldor, IW; Klein, R; Liang, X; McIntyre, MS; Merrill, CL; Patterson, KM; Prescott, JS; Ray, JS; Roller, SG; Yao, X; Young, A; Yuen, J; Collins, JL Discovery of a highly potent, nonabsorbable apical sodium-dependent bile acid transporter inhibitor (GSK2330672) for treatment of type 2 diabetes. J Med Chem56:5094-114 (2013) [PubMed] Article