| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 3A |
|---|
| Ligand | BDBM82561 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_964168 (CHEMBL2394823) |
|---|
| IC50 | 4.9±n/a nM |
|---|
| Citation |  Veinberg, G; Vorona, M; Zvejniece, L; Vilskersts, R; Vavers, E; Liepinsh, E; Kazoka, H; Belyakov, S; Mishnev, A; Kuznecovs, J; Vikainis, S; Orlova, N; Lebedev, A; Ponomaryov, Y; Dambrova, M Synthesis and biological evaluation of 2-(5-methyl-4-phenyl-2-oxopyrrolidin-1-yl)-acetamide stereoisomers as novel positive allosteric modulators of sigma-1 receptor. Bioorg Med Chem21:2764-71 (2013) [PubMed] Article Veinberg, G; Vorona, M; Zvejniece, L; Vilskersts, R; Vavers, E; Liepinsh, E; Kazoka, H; Belyakov, S; Mishnev, A; Kuznecovs, J; Vikainis, S; Orlova, N; Lebedev, A; Ponomaryov, Y; Dambrova, M Synthesis and biological evaluation of 2-(5-methyl-4-phenyl-2-oxopyrrolidin-1-yl)-acetamide stereoisomers as novel positive allosteric modulators of sigma-1 receptor. Bioorg Med Chem21:2764-71 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 3A |
|---|
| Name: | 5-hydroxytryptamine receptor 3A |
|---|
| Synonyms: | 5-HT-3 | 5-HT3 | 5-HT3A | 5-HT3R | 5-hydroxytryptamine receptor 3 (5-HT3) | 5-hydroxytryptamine receptor 3A (5-HT3a) | 5-hydroxytryptamine receptor 3A (5HT3A) | 5HT3A_HUMAN | 5HT3R | HTR3 | HTR3A | Serotonin 3 (5-HT3) receptor | Serotonin 3 receptor (5HT3) | Serotonin receptor 3A | Serotonin-gated ion channel receptor | Serotonin-gated ion channel receptor 3 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55283.27 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P46098 |
|---|
| Residue: | 478 |
|---|
| Sequence: | MLLWVQQALLALLLPTLLAQGEARRSRNTTRPALLRLSDYLLTNYRKGVRPVRDWRKPTT
VSIDVIVYAILNVDEKNQVLTTYIWYRQYWTDEFLQWNPEDFDNITKLSIPTDSIWVPDI
LINEFVDVGKSPNIPYVYIRHQGEVQNYKPLQVVTACSLDIYNFPFDVQNCSLTFTSWLH
TIQDINISLWRLPEKVKSDRSVFMNQGEWELLGVLPYFREFSMESSNYYAEMKFYVVIRR
RPLFYVVSLLLPSIFLMVMDIVGFYLPPNSGERVSFKITLLLGYSVFLIIVSDTLPATAI
GTPLIGVYFVVCMALLVISLAETIFIVRLVHKQDLQQPVPAWLRHLVLERIAWLLCLREQ
STSQRPPATSQATKTDDCSAMGNHCSHMGGPQDFEKSPRDRCSPPPPPREASLAVCGLLQ
ELSSIRQFLEKRDEIREVARDWLRVGSVLDKLLFHIYLLAVLAYSITLVMLWSIWQYA
|
|
|
|---|
| BDBM82561 |
|---|
| n/a |
|---|
| Name | BDBM82561 |
|---|
| Synonyms: | CAS_40796-97-2 | TROPANYL 3,5-DICHLOROBENZOATE | Tropanyl-3,5 dichlorobenzoate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H17Cl2NO2 |
|---|
| Mol. Mass. | 314.207 |
|---|
| SMILES | CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)c1cc(Cl)cc(Cl)c1 |r| |
|---|
| Structure | 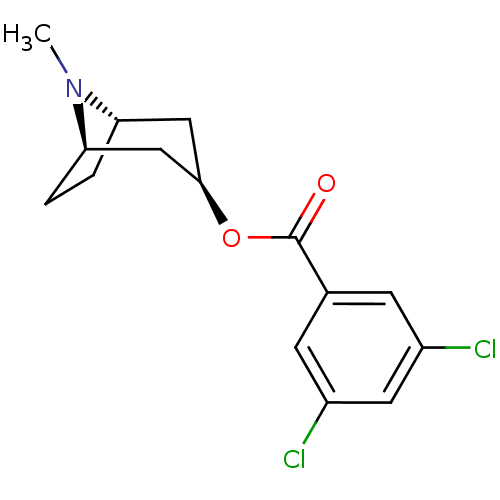
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Veinberg, G; Vorona, M; Zvejniece, L; Vilskersts, R; Vavers, E; Liepinsh, E; Kazoka, H; Belyakov, S; Mishnev, A; Kuznecovs, J; Vikainis, S; Orlova, N; Lebedev, A; Ponomaryov, Y; Dambrova, M Synthesis and biological evaluation of 2-(5-methyl-4-phenyl-2-oxopyrrolidin-1-yl)-acetamide stereoisomers as novel positive allosteric modulators of sigma-1 receptor. Bioorg Med Chem21:2764-71 (2013) [PubMed] Article
Veinberg, G; Vorona, M; Zvejniece, L; Vilskersts, R; Vavers, E; Liepinsh, E; Kazoka, H; Belyakov, S; Mishnev, A; Kuznecovs, J; Vikainis, S; Orlova, N; Lebedev, A; Ponomaryov, Y; Dambrova, M Synthesis and biological evaluation of 2-(5-methyl-4-phenyl-2-oxopyrrolidin-1-yl)-acetamide stereoisomers as novel positive allosteric modulators of sigma-1 receptor. Bioorg Med Chem21:2764-71 (2013) [PubMed] Article