| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Insulin receptor |
|---|
| Ligand | BDBM50436854 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_971485 (CHEMBL2406363) |
|---|
| IC50 | 401±n/a nM |
|---|
| Citation |  Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Insulin receptor |
|---|
| Name: | Insulin receptor |
|---|
| Synonyms: | INSR | INSR protein | INSR_HUMAN | Insulin receptor (IR) | Insulin receptor beta subunit |
|---|
| Type: | Receptor Tyrosine Kinase |
|---|
| Mol. Mass.: | 156322.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06213 |
|---|
| Residue: | 1382 |
|---|
| Sequence: | MATGGRRGAAAAPLLVAVAALLLGAAGHLYPGEVCPGMDIRNNLTRLHELENCSVIEGHL
QILLMFKTRPEDFRDLSFPKLIMITDYLLLFRVYGLESLKDLFPNLTVIRGSRLFFNYAL
VIFEMVHLKELGLYNLMNITRGSVRIEKNNELCYLATIDWSRILDSVEDNYIVLNKDDNE
ECGDICPGTAKGKTNCPATVINGQFVERCWTHSHCQKVCPTICKSHGCTAEGLCCHSECL
GNCSQPDDPTKCVACRNFYLDGRCVETCPPPYYHFQDWRCVNFSFCQDLHHKCKNSRRQG
CHQYVIHNNKCIPECPSGYTMNSSNLLCTPCLGPCPKVCHLLEGEKTIDSVTSAQELRGC
TVINGSLIINIRGGNNLAAELEANLGLIEEISGYLKIRRSYALVSLSFFRKLRLIRGETL
EIGNYSFYALDNQNLRQLWDWSKHNLTITQGKLFFHYNPKLCLSEIHKMEEVSGTKGRQE
RNDIALKTNGDQASCENELLKFSYIRTSFDKILLRWEPYWPPDFRDLLGFMLFYKEAPYQ
NVTEFDGQDACGSNSWTVVDIDPPLRSNDPKSQNHPGWLMRGLKPWTQYAIFVKTLVTFS
DERRTYGAKSDIIYVQTDATNPSVPLDPISVSNSSSQIILKWKPPSDPNGNITHYLVFWE
RQAEDSELFELDYCLKGLKLPSRTWSPPFESEDSQKHNQSEYEDSAGECCSCPKTDSQIL
KELEESSFRKTFEDYLHNVVFVPRKTSSGTGAEDPRPSRKRRSLGDVGNVTVAVPTVAAF
PNTSSTSVPTSPEEHRPFEKVVNKESLVISGLRHFTGYRIELQACNQDTPEERCSVAAYV
SARTMPEAKADDIVGPVTHEIFENNVVHLMWQEPKEPNGLIVLYEVSYRRYGDEELHLCV
SRKHFALERGCRLRGLSPGNYSVRIRATSLAGNGSWTEPTYFYVTDYLDVPSNIAKIIIG
PLIFVFLFSVVIGSIYLFLRKRQPDGPLGPLYASSNPEYLSASDVFPCSVYVPDEWEVSR
EKITLLRELGQGSFGMVYEGNARDIIKGEAETRVAVKTVNESASLRERIEFLNEASVMKG
FTCHHVVRLLGVVSKGQPTLVVMELMAHGDLKSYLRSLRPEAENNPGRPPPTLQEMIQMA
AEIADGMAYLNAKKFVHRDLAARNCMVAHDFTVKIGDFGMTRDIYETDYYRKGGKGLLPV
RWMAPESLKDGVFTTSSDMWSFGVVLWEITSLAEQPYQGLSNEQVLKFVMDGGYLDQPDN
CPERVTDLMRMCWQFNPKMRPTFLEIVNLLKDDLHPSFPEVSFFHSEENKAPESEELEME
FEDMENVPLDRSSHCQREEAGGRDGGSSLGFKRSYEEHIPYTHMNGGKKNGRILTLPRSN
PS
|
|
|
|---|
| BDBM50436854 |
|---|
| n/a |
|---|
| Name | BDBM50436854 |
|---|
| Synonyms: | CHEMBL2403827 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H43N5O3S |
|---|
| Mol. Mass. | 565.77 |
|---|
| SMILES | CCN1CCC(CC1)c1cc(OC(C)C)c(Nc2ncc(C)c(Nc3ccccc3S(=O)(=O)C(C)C)n2)cc1C |
|---|
| Structure | 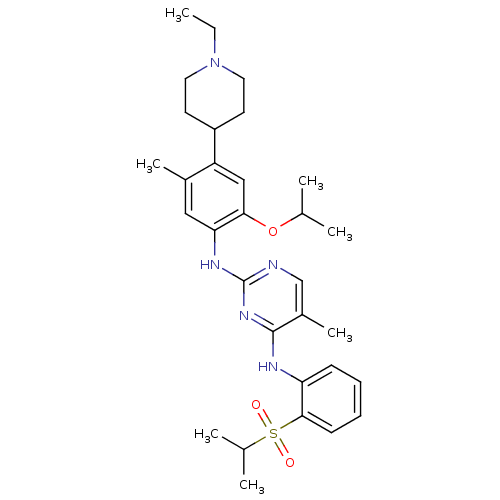
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article
Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article