| Reaction Details |
|---|
 | Report a problem with these data |
| Target | High affinity nerve growth factor receptor |
|---|
| Ligand | BDBM50436850 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_971281 (CHEMBL2405536) |
|---|
| IC50 | 2727±n/a nM |
|---|
| Citation |  Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| High affinity nerve growth factor receptor |
|---|
| Name: | High affinity nerve growth factor receptor |
|---|
| Synonyms: | 2.7.10.1 | MTC | NTRK1 | NTRK1/NTRK2 | NTRK1_HUMAN | Nerve growth factor receptor Trk-A | Neurotrophic tyrosine kinase receptor type 1 | Neurotrophic tyrosine kinase receptor type 1 (TrkA) | Synonyms=MTC | TRK | TRK1-transforming tyrosine kinase protein | TRKA | TRKA GN | Trk-A | Tropomyosin alpha-3 chain/High affinity nerve growth factor receptor | Tropomyosin-related kinase A | Tropomyosin-related kinase A (TrkA) | Tyrosine kinase receptor | Tyrosine kinase receptor (Trk) | Tyrosine kinase receptor A | Tyrosine kinase receptor A (Trk A) | Tyrosine kinase receptor A (Trk-A) | Tyrosine kinase receptor A (TrkA) | gp140trk | p140-TrkA |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 87498.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04629 |
|---|
| Residue: | 796 |
|---|
| Sequence: | MLRGGRRGQLGWHSWAAGPGSLLAWLILASAGAAPCPDACCPHGSSGLRCTRDGALDSLH
HLPGAENLTELYIENQQHLQHLELRDLRGLGELRNLTIVKSGLRFVAPDAFHFTPRLSRL
NLSFNALESLSWKTVQGLSLQELVLSGNPLHCSCALRWLQRWEEEGLGGVPEQKLQCHGQ
GPLAHMPNASCGVPTLKVQVPNASVDVGDDVLLRCQVEGRGLEQAGWILTELEQSATVMK
SGGLPSLGLTLANVTSDLNRKNVTCWAENDVGRAEVSVQVNVSFPASVQLHTAVEMHHWC
IPFSVDGQPAPSLRWLFNGSVLNETSFIFTEFLEPAANETVRHGCLRLNQPTHVNNGNYT
LLAANPFGQASASIMAAFMDNPFEFNPEDPIPVSFSPVDTNSTSGDPVEKKDETPFGVSV
AVGLAVFACLFLSTLLLVLNKCGRRNKFGINRPAVLAPEDGLAMSLHFMTLGGSSLSPTE
GKGSGLQGHIIENPQYFSDACVHHIKRRDIVLKWELGEGAFGKVFLAECHNLLPEQDKML
VAVKALKEASESARQDFQREAELLTMLQHQHIVRFFGVCTEGRPLLMVFEYMRHGDLNRF
LRSHGPDAKLLAGGEDVAPGPLGLGQLLAVASQVAAGMVYLAGLHFVHRDLATRNCLVGQ
GLVVKIGDFGMSRDIYSTDYYRVGGRTMLPIRWMPPESILYRKFTTESDVWSFGVVLWEI
FTYGKQPWYQLSNTEAIDCITQGRELERPRACPPEVYAIMRGCWQREPQQRHSIKDVHAR
LQALAQAPPVYLDVLG
|
|
|
|---|
| BDBM50436850 |
|---|
| n/a |
|---|
| Name | BDBM50436850 |
|---|
| Synonyms: | CERITINIB | CHEMBL2403108 | LDK378 | US10053458, Comparative Example 2 | US10618884, Example 16 | US9809572, Example 16 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H36ClN5O3S |
|---|
| Mol. Mass. | 558.135 |
|---|
| SMILES | CC(C)Oc1cc(C2CCNCC2)c(C)cc1Nc1ncc(Cl)c(Nc2ccccc2S(=O)(=O)C(C)C)n1 |
|---|
| Structure | 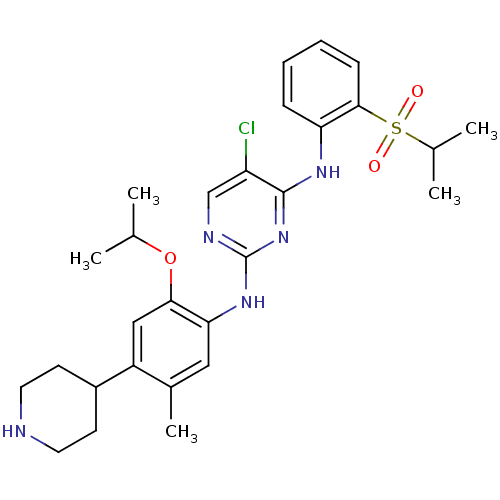
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article
Marsilje, TH; Pei, W; Chen, B; Lu, W; Uno, T; Jin, Y; Jiang, T; Kim, S; Li, N; Warmuth, M; Sarkisova, Y; Sun, F; Steffy, A; Pferdekamper, AC; Li, AG; Joseph, SB; Kim, Y; Liu, B; Tuntland, T; Cui, X; Gray, NS; Steensma, R; Wan, Y; Jiang, J; Chopiuk, G; Li, J; Gordon, WP; Richmond, W; Johnson, K; Chang, J; Groessl, T; He, YQ; Phimister, A; Aycinena, A; Lee, CC; Bursulaya, B; Karanewsky, DS; Seidel, HM; Harris, JL; Michellys, PY Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase J Med Chem56:5675-90 (2014) [PubMed] Article