| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase receptor TYRO3 |
|---|
| Ligand | BDBM50442259 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_991999 (CHEMBL2447154) |
|---|
| IC50 | >1000±n/a nM |
|---|
| Citation |  Zhang, D; Zhang, X; Ai, J; Zhai, Y; Liang, Z; Wang, Y; Chen, Y; Li, C; Zhao, F; Jiang, H; Geng, M; Luo, C; Liu, H Synthesis and biological evaluation of 2-amino-5-aryl-3-benzylthiopyridine scaffold based potent c-Met inhibitors. Bioorg Med Chem21:6804-20 (2013) [PubMed] Article Zhang, D; Zhang, X; Ai, J; Zhai, Y; Liang, Z; Wang, Y; Chen, Y; Li, C; Zhao, F; Jiang, H; Geng, M; Luo, C; Liu, H Synthesis and biological evaluation of 2-amino-5-aryl-3-benzylthiopyridine scaffold based potent c-Met inhibitors. Bioorg Med Chem21:6804-20 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase receptor TYRO3 |
|---|
| Name: | Tyrosine-protein kinase receptor TYRO3 |
|---|
| Synonyms: | BYK | DTK | RSE | SKY | TIF | TYRO3 | TYRO3_HUMAN | Tyrosine-protein kinase SKY |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 96894.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q06418 |
|---|
| Residue: | 890 |
|---|
| Sequence: | MALRRSMGRPGLPPLPLPPPPRLGLLLAALASLLLPESAAAGLKLMGAPVKLTVSQGQPV
KLNCSVEGMEEPDIQWVKDGAVVQNLDQLYIPVSEQHWIGFLSLKSVERSDAGRYWCQVE
DGGETEISQPVWLTVEGVPFFTVEPKDLAVPPNAPFQLSCEAVGPPEPVTIVWWRGTTKI
GGPAPSPSVLNVTGVTQSTMFSCEAHNLKGLASSRTATVHLQALPAAPFNITVTKLSSSN
ASVAWMPGADGRALLQSCTVQVTQAPGGWEVLAVVVPVPPFTCLLRDLVPATNYSLRVRC
ANALGPSPYADWVPFQTKGLAPASAPQNLHAIRTDSGLILEWEEVIPEAPLEGPLGPYKL
SWVQDNGTQDELTVEGTRANLTGWDPQKDLIVRVCVSNAVGCGPWSQPLVVSSHDRAGQQ
GPPHSRTSWVPVVLGVLTALVTAAALALILLRKRRKETRFGQAFDSVMARGEPAVHFRAA
RSFNRERPERIEATLDSLGISDELKEKLEDVLIPEQQFTLGRMLGKGEFGSVREAQLKQE
DGSFVKVAVKMLKADIIASSDIEEFLREAACMKEFDHPHVAKLVGVSLRSRAKGRLPIPM
VILPFMKHGDLHAFLLASRIGENPFNLPLQTLIRFMVDIACGMEYLSSRNFIHRDLAARN
CMLAEDMTVCVADFGLSRKIYSGDYYRQGCASKLPVKWLALESLADNLYTVQSDVWAFGV
TMWEIMTRGQTPYAGIENAEIYNYLIGGNRLKQPPECMEDVYDLMYQCWSADPKQRPSFT
CLRMELENILGQLSVLSASQDPLYINIERAEEPTAGGSLELPGRDQPYSGAGDGSGMGAV
GGTPSDCRYILTPGGLAEQPGQAEHQPESPLNETQRLLLLQQGLLPHSSC
|
|
|
|---|
| BDBM50442259 |
|---|
| n/a |
|---|
| Name | BDBM50442259 |
|---|
| Synonyms: | CHEMBL2442124 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H22Cl2FN5S |
|---|
| Mol. Mass. | 466.402 |
|---|
| SMILES | C[C@@H](Sc1cc(cnc1N)-c1cnn(c1)C1CCNCC1)c1c(Cl)ccc(F)c1Cl |r| |
|---|
| Structure | 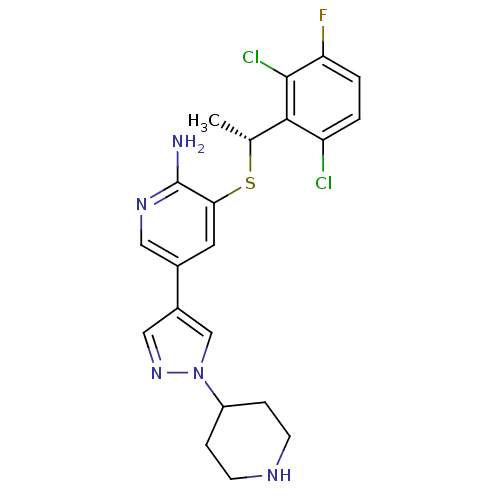
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, D; Zhang, X; Ai, J; Zhai, Y; Liang, Z; Wang, Y; Chen, Y; Li, C; Zhao, F; Jiang, H; Geng, M; Luo, C; Liu, H Synthesis and biological evaluation of 2-amino-5-aryl-3-benzylthiopyridine scaffold based potent c-Met inhibitors. Bioorg Med Chem21:6804-20 (2013) [PubMed] Article
Zhang, D; Zhang, X; Ai, J; Zhai, Y; Liang, Z; Wang, Y; Chen, Y; Li, C; Zhao, F; Jiang, H; Geng, M; Luo, C; Liu, H Synthesis and biological evaluation of 2-amino-5-aryl-3-benzylthiopyridine scaffold based potent c-Met inhibitors. Bioorg Med Chem21:6804-20 (2013) [PubMed] Article