| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2X purinoceptor 1 |
|---|
| Ligand | BDBM50102295 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1280008 (CHEMBL3096835) |
|---|
| IC50 | 43±n/a nM |
|---|
| Citation |  Cho, JH; Jung, KY; Jung, Y; Kim, MH; Ko, H; Park, CS; Kim, YC Design and synthesis of potent and selective P2X3 receptor antagonists derived from PPADS as potential pain modulators. Eur J Med Chem70:811-30 (2013) [PubMed] Article Cho, JH; Jung, KY; Jung, Y; Kim, MH; Ko, H; Park, CS; Kim, YC Design and synthesis of potent and selective P2X3 receptor antagonists derived from PPADS as potential pain modulators. Eur J Med Chem70:811-30 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2X purinoceptor 1 |
|---|
| Name: | P2X purinoceptor 1 |
|---|
| Synonyms: | ATP receptor | P2RX1_RAT | P2X1 | P2rx1 | Purinergic receptor | Purinergic, P2X1 | RP-2 protein |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44969.95 |
|---|
| Organism: | RAT |
|---|
| Description: | Purinergic, P2X1 0 RAT::P47824 |
|---|
| Residue: | 399 |
|---|
| Sequence: | MARRLQDELSAFFFEYDTPRMVLVRNKKVGVIFRLIQLVVLVYVIGWVFVYEKGYQTSSD
LISSVSVKLKGLAVTQLQGLGPQVWDVADYVFPAHGDSSFVVMTNFIVTPQQTQGHCAEN
PEGGICQDDSGCTPGKAERKAQGIRTGNCVPFNGTVKTCEIFGWCPVEVDDKIPSPALLR
EAENFTLFIKNSISFPRFKVNRRNLVEEVNGTYMKKCLYHKIQHPLCPVFNLGYVVRESG
QDFRSLAEKGGVVGITIDWKCDLDWHVRHCKPIYQFHGLYGEKNLSPGFNFRFARHFVQN
GTNRRHLFKVFGIHFDILVDGKAGKFDIIPTMTTIGSGIGIFGVATVLCDLLLLHILPKR
HYYKQKKFKYAEDMGPGEGEHDPVATSSTLGLQENMRTS
|
|
|
|---|
| BDBM50102295 |
|---|
| n/a |
|---|
| Name | BDBM50102295 |
|---|
| Synonyms: | 4-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethyl-pyridin-2-ylazo)-benzoic acid | CHEMBL119235 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H14N3O8P |
|---|
| Mol. Mass. | 395.2607 |
|---|
| SMILES | Cc1nc(N=Nc2ccc(cc2)C(O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:5.5| |
|---|
| Structure | 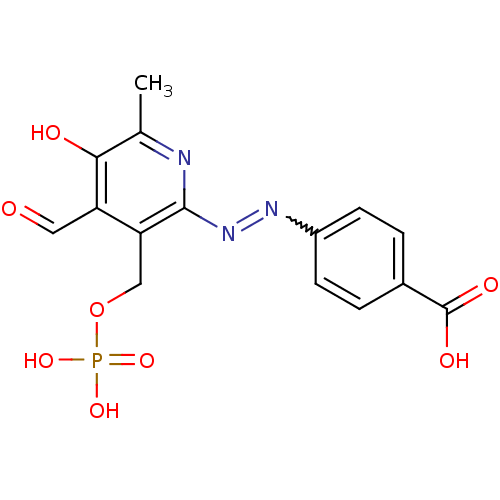
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cho, JH; Jung, KY; Jung, Y; Kim, MH; Ko, H; Park, CS; Kim, YC Design and synthesis of potent and selective P2X3 receptor antagonists derived from PPADS as potential pain modulators. Eur J Med Chem70:811-30 (2013) [PubMed] Article
Cho, JH; Jung, KY; Jung, Y; Kim, MH; Ko, H; Park, CS; Kim, YC Design and synthesis of potent and selective P2X3 receptor antagonists derived from PPADS as potential pain modulators. Eur J Med Chem70:811-30 (2013) [PubMed] Article