| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutamate receptor ionotropic, NMDA 2B |
|---|
| Ligand | BDBM50083351 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1283718 (CHEMBL3108298) |
|---|
| IC50 | 20±n/a nM |
|---|
| Citation |  Gitto, R; De Luca, L; Ferro, S; Russo, E; De Sarro, G; Chisari, M; Ciranna, L; Alvarez-Builla, J; Alajarin, R; Buemi, MR; Chimirri, A Synthesis, modelling and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-d-aspartate receptors. Bioorg Med Chem22:1040-8 (2014) [PubMed] Article Gitto, R; De Luca, L; Ferro, S; Russo, E; De Sarro, G; Chisari, M; Ciranna, L; Alvarez-Builla, J; Alajarin, R; Buemi, MR; Chimirri, A Synthesis, modelling and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-d-aspartate receptors. Bioorg Med Chem22:1040-8 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutamate receptor ionotropic, NMDA 2B |
|---|
| Name: | Glutamate receptor ionotropic, NMDA 2B |
|---|
| Synonyms: | GluN2B | Glutamate [NMDA] receptor subunit epsilon 2 | Grin2b | N-methyl D-aspartate receptor subtype 2B | NMDA receptor subunit N2B (GluN2B) | NMDAR2B | NMDE2_RAT | NR2B |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 166077.66 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | Q00960 |
|---|
| Residue: | 1482 |
|---|
| Sequence: | MKPSAECCSPKFWLVLAVLAVSGSKARSQKSPPSIGIAVILVGTSDEVAIKDAHEKDDFH
HLSVVPRVELVAMNETDPKSIITRICDLMSDRKIQGVVFADDTDQEAIAQILDFISAQTL
TPILGIHGGSSMIMADKDESSMFFQFGPSIEQQASVMLNIMEEYDWYIFSIVTTYFPGYQ
DFVNKIRSTIENSFVGWELEEVLLLDMSLDDGDSKIQNQLKKLQSPIILLYCTKEEATYI
FEVANSVGLTGYGYTWIVPSLVAGDTDTVPSEFPTGLISVSYDEWDYGLPARVRDGIAII
TTAASDMLSEHSFIPEPKSSCYNTHEKRIYQSNMLNRYLINVTFEGRNLSFSEDGYQMHP
KLVIILLNKERKWERVGKWKDKSLQMKYYVWPRMCPETEEQEDDHLSIVTLEEAPFVIVE
SVDPLSGTCMRNTVPCQKRIISENKTDEEPGYIKKCCKGFCIDILKKISKSVKFTYDLYL
VTNGKHGKKINGTWNGMIGEVVMKRAYMAVGSLTINEERSEVVDFSVPFIETGISVMVSR
SNGTVSPSAFLEPFSADVWVMMFVMLLIVSAVAVFVFEYFSPVGYNRCLADGREPGGPSF
TIGKAIWLLWGLVFNNSVPVQNPKGTTSKIMVSVWAFFAVIFLASYTANLAAFMIQEEYV
DQVSGLSDKKFQRPNDFSPPFRFGTVPNGSTERNIRNNYAEMHAYMGKFNQRGVDDALLS
LKTGKLDAFIYDAAVLNYMAGRDEGCKLVTIGSGKVFASTGYGIAIQKDSGWKRQVDLAI
LQLFGDGEMEELEALWLTGICHNEKNEVMSSQLDIDNMAGVFYMLGAAMALSLITFICEH
LFYWQFRHCFMGVCSGKPGMVFSISRGIYSCIHGVAIEERQSVMNSPTATMNNTHSNILR
LLRTAKNMANLSGVNGSPQSALDFIRRESSVYDISEHRRSFTHSDCKSYNNPPCEENLFS
DYISEVERTFGNLQLKDSNVYQDHYHHHHRPHSIGSTSSIDGLYDCDNPPFTTQPRSISK
KPLDIGLPSSKHSQLSDLYGKFSFKSDRYSGHDDLIRSDVSDISTHTVTYGNIEGNAAKR
RKQQYKDSLKKRPASAKSRREFDEIELAYRRRPPRSPDHKRYFRDKEGLRDFYLDQFRTK
ENSPHWEHVDLTDIYKERSDDFKRDSVSGGGPCTNRSHLKHGTGEKHGVVGGVPAPWEKN
LTNVDWEDRSGGNFCRSCPSKLHNYSSTVAGQNSGRQACIRCEACKKAGNLYDISEDNSL
QELDQPAAPVAVTSNASSTKYPQSPTNSKAQKKNRNKLRRQHSYDTFVDLQKEEAALAPR
SVSLKDKGRFMDGSPYAHMFEMPAGESSFANKSSVPTAGHHHNNPGSGYMLSKSLYPDRV
TQNPFIPTFGDDQCLLHGSKSYFFRQPTVAGASKTRPDFRALVTNKPVVVTLHGAVPGRF
QKDICIGNQSNPCVPNNKNPRAFNGSSNGHVYEKLSSIESDV
|
|
|
|---|
| BDBM50083351 |
|---|
| n/a |
|---|
| Name | BDBM50083351 |
|---|
| Synonyms: | (+/-)-[2-(4-benzylpiperidino)-1-(4-hydroxyphenyl)-1-propanol] | -(2-(4-benzylpiperidin-1-yl)-1-hydroxypropyl)phenol | 4-(2-(4-benzylpiperidin-1-yl)-1-hydroxypropyl)phenol | 4-[2-(4-Benzyl-piperidin-1-yl)-1-hydroxy-propyl]-phenol | CHEMBL305187 | ChEMBL_104385 | IFENPRODIL |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H27NO2 |
|---|
| Mol. Mass. | 325.4446 |
|---|
| SMILES | CC(C(O)c1ccc(O)cc1)N1CCC(Cc2ccccc2)CC1 |
|---|
| Structure | 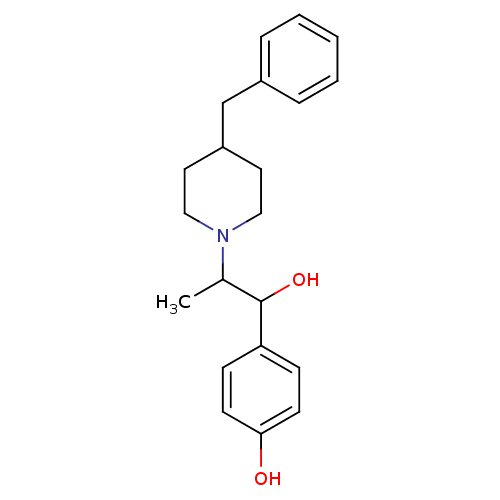
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gitto, R; De Luca, L; Ferro, S; Russo, E; De Sarro, G; Chisari, M; Ciranna, L; Alvarez-Builla, J; Alajarin, R; Buemi, MR; Chimirri, A Synthesis, modelling and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-d-aspartate receptors. Bioorg Med Chem22:1040-8 (2014) [PubMed] Article
Gitto, R; De Luca, L; Ferro, S; Russo, E; De Sarro, G; Chisari, M; Ciranna, L; Alvarez-Builla, J; Alajarin, R; Buemi, MR; Chimirri, A Synthesis, modelling and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-d-aspartate receptors. Bioorg Med Chem22:1040-8 (2014) [PubMed] Article