| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50003477 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1335568 (CHEMBL3239423) |
|---|
| IC50 | <100±n/a nM |
|---|
| Citation |  Phillips, DP; Gao, W; Yang, Y; Zhang, G; Lerario, IK; Lau, TL; Jiang, J; Wang, X; Nguyen, DG; Bhat, BG; Trotter, C; Sullivan, H; Welzel, G; Landry, J; Chen, Y; Joseph, SB; Li, C; Gordon, WP; Richmond, W; Johnson, K; Bretz, A; Bursulaya, B; Pan, S; McNamara, P; Seidel, HM Discovery of trifluoromethyl(pyrimidin-2-yl)azetidine-2-carboxamides as potent, orally bioavailable TGR5 (GPBAR1) agonists: structure-activity relationships, lead optimization, and chronic in vivo efficacy. J Med Chem57:3263-82 (2014) [PubMed] Article Phillips, DP; Gao, W; Yang, Y; Zhang, G; Lerario, IK; Lau, TL; Jiang, J; Wang, X; Nguyen, DG; Bhat, BG; Trotter, C; Sullivan, H; Welzel, G; Landry, J; Chen, Y; Joseph, SB; Li, C; Gordon, WP; Richmond, W; Johnson, K; Bretz, A; Bursulaya, B; Pan, S; McNamara, P; Seidel, HM Discovery of trifluoromethyl(pyrimidin-2-yl)azetidine-2-carboxamides as potent, orally bioavailable TGR5 (GPBAR1) agonists: structure-activity relationships, lead optimization, and chronic in vivo efficacy. J Med Chem57:3263-82 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50003477 |
|---|
| n/a |
|---|
| Name | BDBM50003477 |
|---|
| Synonyms: | CHEMBL3234569 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H28F3N7O |
|---|
| Mol. Mass. | 487.5206 |
|---|
| SMILES | FC(F)(F)c1nc(cc(n1)N1CCC[C@H](C1)C(=O)NCCc1ccc(cc1)C#N)N1CCNCC1 |r| |
|---|
| Structure | 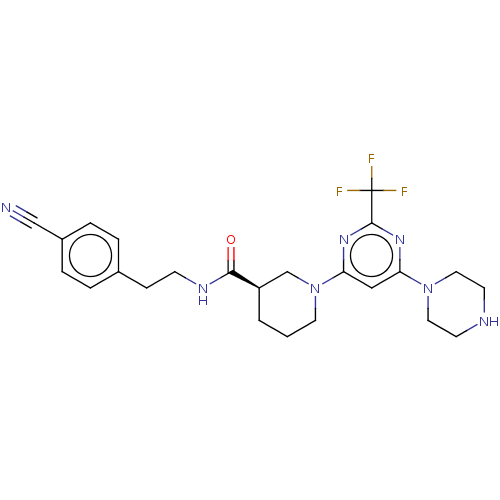
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Phillips, DP; Gao, W; Yang, Y; Zhang, G; Lerario, IK; Lau, TL; Jiang, J; Wang, X; Nguyen, DG; Bhat, BG; Trotter, C; Sullivan, H; Welzel, G; Landry, J; Chen, Y; Joseph, SB; Li, C; Gordon, WP; Richmond, W; Johnson, K; Bretz, A; Bursulaya, B; Pan, S; McNamara, P; Seidel, HM Discovery of trifluoromethyl(pyrimidin-2-yl)azetidine-2-carboxamides as potent, orally bioavailable TGR5 (GPBAR1) agonists: structure-activity relationships, lead optimization, and chronic in vivo efficacy. J Med Chem57:3263-82 (2014) [PubMed] Article
Phillips, DP; Gao, W; Yang, Y; Zhang, G; Lerario, IK; Lau, TL; Jiang, J; Wang, X; Nguyen, DG; Bhat, BG; Trotter, C; Sullivan, H; Welzel, G; Landry, J; Chen, Y; Joseph, SB; Li, C; Gordon, WP; Richmond, W; Johnson, K; Bretz, A; Bursulaya, B; Pan, S; McNamara, P; Seidel, HM Discovery of trifluoromethyl(pyrimidin-2-yl)azetidine-2-carboxamides as potent, orally bioavailable TGR5 (GPBAR1) agonists: structure-activity relationships, lead optimization, and chronic in vivo efficacy. J Med Chem57:3263-82 (2014) [PubMed] Article