| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase |
|---|
| Ligand | BDBM50155838 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1336165 (CHEMBL3241125) |
|---|
| Ki | 0.023000±n/a nM |
|---|
| Citation |  Kallinen, A; Todorov, B; Kallionp��, R; B�ck, S; Sarparanta, M; Raki, M; Garc�a-Horsman, JA; Bergstr�m, KA; Wall�n, EA; M�nnist�, PT; Airaksinen, AJ Synthesis and biological evaluation of novel (123)I-labeled 4-(4-iodophenyl)butanoyl-L-prolyl-(2S)-pyrrolidines for imaging prolyl oligopeptidase in vivo. Eur J Med Chem79:436-45 (2014) [PubMed] Article Kallinen, A; Todorov, B; Kallionp��, R; B�ck, S; Sarparanta, M; Raki, M; Garc�a-Horsman, JA; Bergstr�m, KA; Wall�n, EA; M�nnist�, PT; Airaksinen, AJ Synthesis and biological evaluation of novel (123)I-labeled 4-(4-iodophenyl)butanoyl-L-prolyl-(2S)-pyrrolidines for imaging prolyl oligopeptidase in vivo. Eur J Med Chem79:436-45 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase |
|---|
| Name: | Prolyl endopeptidase |
|---|
| Synonyms: | PE | PEP | POP | PPCE_HUMAN | PREP | Post-proline cleaving enzyme | Prolyl oligopeptidase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 80688.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P48147 |
|---|
| Residue: | 710 |
|---|
| Sequence: | MLSLQYPDVYRDETAVQDYHGHKICDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIR
GLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILS
DDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTH
DGKGMFYNSYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAEL
SDDGRYVLLSIREGCDPVNRLWYCDLQQESSGIAGILKWVKLIDNFEGEYDYVTNEGTVF
TFKTNRQSPNYRVINIDFRDPEESKWKVLVPEHEKDVLEWIACVRSNFLVLCYLHDVKNI
LQLHDLTTGALLKTFPLDVGSIVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPR
VFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISI
TPNYSVSRLIFVRHMGGILAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKE
GYTSPKRLTINGGSNGGLLVAACANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYG
CSDSKQHFEWLVKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIV
GRSRKQSNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNVDWIP
|
|
|
|---|
| BDBM50155838 |
|---|
| n/a |
|---|
| Name | BDBM50155838 |
|---|
| Synonyms: | (S)-1-((S)-1-(4-phenylbutanoyl)pyrrolidine-2-carbonyl)pyrrolidine-2-carbonitrile | (S)-1-[(S)-1-(4-Phenyl-butyryl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonitrile | CHEMBL189620 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H25N3O2 |
|---|
| Mol. Mass. | 339.4314 |
|---|
| SMILES | O=C(CCCc1ccccc1)N1CCC[C@H]1C(=O)N1CCC[C@H]1C#N |
|---|
| Structure | 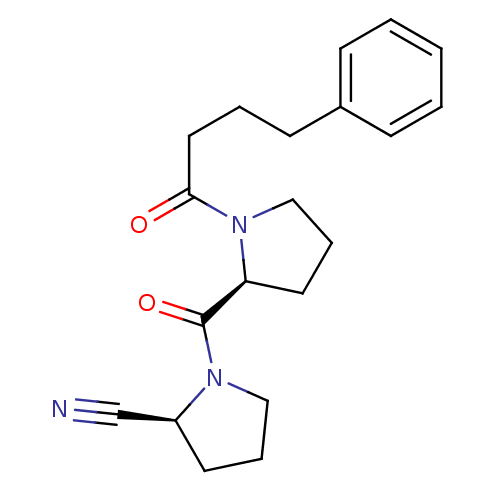
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kallinen, A; Todorov, B; Kallionp��, R; B�ck, S; Sarparanta, M; Raki, M; Garc�a-Horsman, JA; Bergstr�m, KA; Wall�n, EA; M�nnist�, PT; Airaksinen, AJ Synthesis and biological evaluation of novel (123)I-labeled 4-(4-iodophenyl)butanoyl-L-prolyl-(2S)-pyrrolidines for imaging prolyl oligopeptidase in vivo. Eur J Med Chem79:436-45 (2014) [PubMed] Article
Kallinen, A; Todorov, B; Kallionp��, R; B�ck, S; Sarparanta, M; Raki, M; Garc�a-Horsman, JA; Bergstr�m, KA; Wall�n, EA; M�nnist�, PT; Airaksinen, AJ Synthesis and biological evaluation of novel (123)I-labeled 4-(4-iodophenyl)butanoyl-L-prolyl-(2S)-pyrrolidines for imaging prolyl oligopeptidase in vivo. Eur J Med Chem79:436-45 (2014) [PubMed] Article