| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50448936 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1336778 (CHEMBL3241317) |
|---|
| IC50 | >27000±n/a nM |
|---|
| Citation |  Gonzalez, AZ; Li, Z; Beck, HP; Canon, J; Chen, A; Chow, D; Duquette, J; Eksterowicz, J; Fox, BM; Fu, J; Huang, X; Houze, J; Jin, L; Li, Y; Ling, Y; Lo, MC; Long, AM; McGee, LR; McIntosh, J; Oliner, JD; Osgood, T; Rew, Y; Saiki, AY; Shaffer, P; Wortman, S; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Olson, SH; Sun, D; Medina, JC Novel inhibitors of the MDM2-p53 interaction featuring hydrogen bond acceptors as carboxylic acid isosteres. J Med Chem57:2963-88 (2014) [PubMed] Article Gonzalez, AZ; Li, Z; Beck, HP; Canon, J; Chen, A; Chow, D; Duquette, J; Eksterowicz, J; Fox, BM; Fu, J; Huang, X; Houze, J; Jin, L; Li, Y; Ling, Y; Lo, MC; Long, AM; McGee, LR; McIntosh, J; Oliner, JD; Osgood, T; Rew, Y; Saiki, AY; Shaffer, P; Wortman, S; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Olson, SH; Sun, D; Medina, JC Novel inhibitors of the MDM2-p53 interaction featuring hydrogen bond acceptors as carboxylic acid isosteres. J Med Chem57:2963-88 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50448936 |
|---|
| n/a |
|---|
| Name | BDBM50448936 |
|---|
| Synonyms: | CHEMBL3125527 | US9296736, 342 | US9593129, Example 342 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H35Cl2NO5S |
|---|
| Mol. Mass. | 568.552 |
|---|
| SMILES | CC[C@@H](CS(=O)(=O)C(C)(C)C)N1[C@@H]([C@H](C[C@](C)(CC(O)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| |
|---|
| Structure | 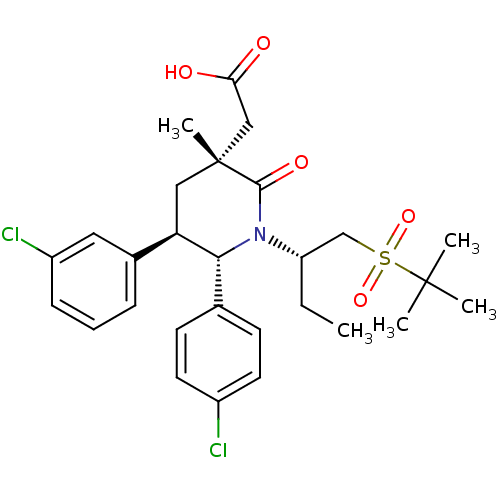
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gonzalez, AZ; Li, Z; Beck, HP; Canon, J; Chen, A; Chow, D; Duquette, J; Eksterowicz, J; Fox, BM; Fu, J; Huang, X; Houze, J; Jin, L; Li, Y; Ling, Y; Lo, MC; Long, AM; McGee, LR; McIntosh, J; Oliner, JD; Osgood, T; Rew, Y; Saiki, AY; Shaffer, P; Wortman, S; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Olson, SH; Sun, D; Medina, JC Novel inhibitors of the MDM2-p53 interaction featuring hydrogen bond acceptors as carboxylic acid isosteres. J Med Chem57:2963-88 (2014) [PubMed] Article
Gonzalez, AZ; Li, Z; Beck, HP; Canon, J; Chen, A; Chow, D; Duquette, J; Eksterowicz, J; Fox, BM; Fu, J; Huang, X; Houze, J; Jin, L; Li, Y; Ling, Y; Lo, MC; Long, AM; McGee, LR; McIntosh, J; Oliner, JD; Osgood, T; Rew, Y; Saiki, AY; Shaffer, P; Wortman, S; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Olson, SH; Sun, D; Medina, JC Novel inhibitors of the MDM2-p53 interaction featuring hydrogen bond acceptors as carboxylic acid isosteres. J Med Chem57:2963-88 (2014) [PubMed] Article