| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Thyroid hormone receptor beta |
|---|
| Ligand | BDBM18869 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1351953 (CHEMBL3266217) |
|---|
| EC50 | 23±n/a nM |
|---|
| Citation |  Kelly, MJ; Pietranico-Cole, S; Larigan, JD; Haynes, NE; Reynolds, CH; Scott, N; Vermeulen, J; Dvorozniak, M; Conde-Knape, K; Huang, KS; So, SS; Thakkar, K; Qian, Y; Banner, B; Mennona, F; Danzi, S; Klein, I; Taub, R; Tilley, J Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a Highly Selective Thyroid Hormone Receptor� agonist in clinical trials for the treatment of dyslipidemia. J Med Chem57:3912-23 (2014) [PubMed] Article Kelly, MJ; Pietranico-Cole, S; Larigan, JD; Haynes, NE; Reynolds, CH; Scott, N; Vermeulen, J; Dvorozniak, M; Conde-Knape, K; Huang, KS; So, SS; Thakkar, K; Qian, Y; Banner, B; Mennona, F; Danzi, S; Klein, I; Taub, R; Tilley, J Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a Highly Selective Thyroid Hormone Receptor� agonist in clinical trials for the treatment of dyslipidemia. J Med Chem57:3912-23 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Thyroid hormone receptor beta |
|---|
| Name: | Thyroid hormone receptor beta |
|---|
| Synonyms: | ERBA2 | NR1A2 | Nuclear receptor subfamily 1 group A member 2 | THB_HUMAN | THR1 | THRB | c-erbA-2 | c-erbA-beta |
|---|
| Type: | Receptor |
|---|
| Mol. Mass.: | 52793.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant hThR was obtained from nuclear extracts from SF9 cells infected with baculovirus vectors encoding for ThRbeta 1. |
|---|
| Residue: | 461 |
|---|
| Sequence: | MTPNSMTENGLTAWDKPKHCPDREHDWKLVGMSEACLHRKSHSERRSTLKNEQSSPHLIQ
TTWTSSIFHLDHDDVNDQSVSSAQTFQTEEKKCKGYIPSYLDKDELCVVCGDKATGYHYR
CITCEGCKGFFRRTIQKNLHPSYSCKYEGKCVIDKVTRNQCQECRFKKCIYVGMATDLVL
DDSKRLAKRKLIEENREKRRREELQKSIGHKPEPTDEEWELIKTVTEAHVATNAQGSHWK
QKRKFLPEDIGQAPIVNAPEGGKVDLEAFSHFTKIITPAITRVVDFAKKLPMFCELPCED
QIILLKGCCMEIMSLRAAVRYDPESETLTLNGEMAVTRGQLKNGGLGVVSDAIFDLGMSL
SSFNLDDTEVALLQAVLLMSSDRPGLACVERIEKYQDSFLLAFEHYINYRKHHVTHFWPK
LLMKVTDLRMIGACHASRFLHMKVECPTELFPPLFLEVFED
|
|
|
|---|
| BDBM18869 |
|---|
| n/a |
|---|
| Name | BDBM18869 |
|---|
| Synonyms: | 2-{3,5-dichloro-4-[4-hydroxy-3-(propan-2-yl)phenoxy]phenyl}acetic acid | CHEMBL41036 | KB-141 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H16Cl2O4 |
|---|
| Mol. Mass. | 355.213 |
|---|
| SMILES | CC(C)c1cc(Oc2c(Cl)cc(CC(O)=O)cc2Cl)ccc1O |
|---|
| Structure | 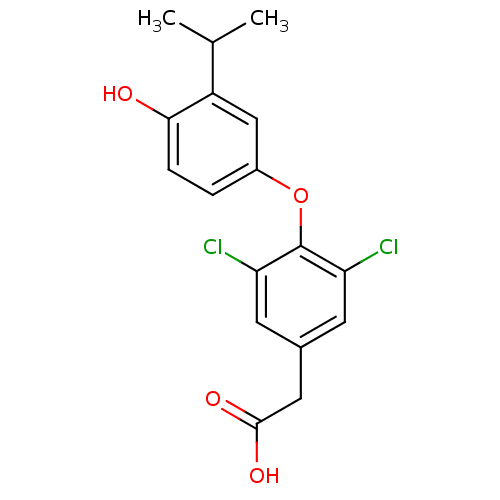
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kelly, MJ; Pietranico-Cole, S; Larigan, JD; Haynes, NE; Reynolds, CH; Scott, N; Vermeulen, J; Dvorozniak, M; Conde-Knape, K; Huang, KS; So, SS; Thakkar, K; Qian, Y; Banner, B; Mennona, F; Danzi, S; Klein, I; Taub, R; Tilley, J Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a Highly Selective Thyroid Hormone Receptor� agonist in clinical trials for the treatment of dyslipidemia. J Med Chem57:3912-23 (2014) [PubMed] Article
Kelly, MJ; Pietranico-Cole, S; Larigan, JD; Haynes, NE; Reynolds, CH; Scott, N; Vermeulen, J; Dvorozniak, M; Conde-Knape, K; Huang, KS; So, SS; Thakkar, K; Qian, Y; Banner, B; Mennona, F; Danzi, S; Klein, I; Taub, R; Tilley, J Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a Highly Selective Thyroid Hormone Receptor� agonist in clinical trials for the treatment of dyslipidemia. J Med Chem57:3912-23 (2014) [PubMed] Article