| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fatty-acid amide hydrolase 1 |
|---|
| Ligand | BDBM50021344 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1366315 (CHEMBL3295961) |
|---|
| IC50 | 13±n/a nM |
|---|
| Citation |  Chobanian, HR; Guo, Y; Liu, P; Chioda, MD; Fung, S; Lanza, TJ; Chang, L; Bakshi, RK; Dellureficio, JP; Hong, Q; McLaughlin, M; Belyk, KM; Krska, SW; Makarewicz, AK; Martel, EJ; Leone, JF; Frey, L; Karanam, B; Madeira, M; Alvaro, R; Shuman, J; Salituro, G; Terebetski, JL; Jochnowitz, N; Mistry, S; McGowan, E; Hajdu, R; Rosenbach, M; Abbadie, C; Alexander, JP; Shiao, LL; Sullivan, KM; Nargund, RP; Wyvratt, MJ; Lin, LS; DeVita, RJ Discovery of MK-4409, a Novel Oxazole FAAH Inhibitor for the Treatment of Inflammatory and Neuropathic Pain. ACS Med Chem Lett5:717-21 (2014) [PubMed] Article Chobanian, HR; Guo, Y; Liu, P; Chioda, MD; Fung, S; Lanza, TJ; Chang, L; Bakshi, RK; Dellureficio, JP; Hong, Q; McLaughlin, M; Belyk, KM; Krska, SW; Makarewicz, AK; Martel, EJ; Leone, JF; Frey, L; Karanam, B; Madeira, M; Alvaro, R; Shuman, J; Salituro, G; Terebetski, JL; Jochnowitz, N; Mistry, S; McGowan, E; Hajdu, R; Rosenbach, M; Abbadie, C; Alexander, JP; Shiao, LL; Sullivan, KM; Nargund, RP; Wyvratt, MJ; Lin, LS; DeVita, RJ Discovery of MK-4409, a Novel Oxazole FAAH Inhibitor for the Treatment of Inflammatory and Neuropathic Pain. ACS Med Chem Lett5:717-21 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Fatty-acid amide hydrolase 1 |
|---|
| Name: | Fatty-acid amide hydrolase 1 |
|---|
| Synonyms: | Anandamide amidohydrolase | Anandamide amidohydrolase 1 | FAAH | FAAH1 | FAAH1_HUMAN | Fatty Acid Amide Hydrolase (FAAH) | Fatty-acid amide hydrolase (FAAH) | Fatty-acid amide hydrolase 1 | Oleamide hydrolase 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 63071.19 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O00519 |
|---|
| Residue: | 579 |
|---|
| Sequence: | MVQYELWAALPGASGVALACCFVAAAVALRWSGRRTARGAVVRARQRQRAGLENMDRAAQ
RFRLQNPDLDSEALLALPLPQLVQKLHSRELAPEAVLFTYVGKAWEVNKGTNCVTSYLAD
CETQLSQAPRQGLLYGVPVSLKECFTYKGQDSTLGLSLNEGVPAECDSVVVHVLKLQGAV
PFVHTNVPQSMFSYDCSNPLFGQTVNPWKSSKSPGGSSGGEGALIGSGGSPLGLGTDIGG
SIRFPSSFCGICGLKPTGNRLSKSGLKGCVYGQEAVRLSVGPMARDVESLALCLRALLCE
DMFRLDPTVPPLPFREEVYTSSQPLRVGYYETDNYTMPSPAMRRAVLETKQSLEAAGHTL
VPFLPSNIPHALETLSTGGLFSDGGHTFLQNFKGDFVDPCLGDLVSILKLPQWLKGLLAF
LVKPLLPRLSAFLSNMKSRSAGKLWELQHEIEVYRKTVIAQWRALDLDVVLTPMLAPALD
LNAPGRATGAVSYTMLYNCLDFPAGVVPVTTVTAEDEAQMEHYRGYFGDIWDKMLQKGMK
KSVGLPVAVQCVALPWQEELCLRFMREVERLMTPEKQSS
|
|
|
|---|
| BDBM50021344 |
|---|
| n/a |
|---|
| Name | BDBM50021344 |
|---|
| Synonyms: | CHEMBL3287929 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H14ClN5OS |
|---|
| Mol. Mass. | 431.897 |
|---|
| SMILES | Clc1ccc(Sc2cn(nc2-c2ccc(cc2)-c2ncon2)-c2cccnc2)cc1 |
|---|
| Structure | 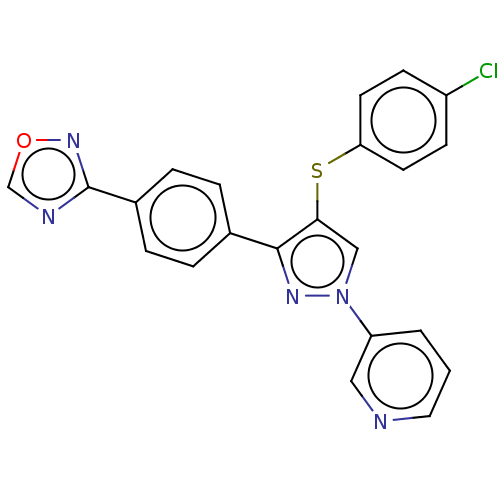
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chobanian, HR; Guo, Y; Liu, P; Chioda, MD; Fung, S; Lanza, TJ; Chang, L; Bakshi, RK; Dellureficio, JP; Hong, Q; McLaughlin, M; Belyk, KM; Krska, SW; Makarewicz, AK; Martel, EJ; Leone, JF; Frey, L; Karanam, B; Madeira, M; Alvaro, R; Shuman, J; Salituro, G; Terebetski, JL; Jochnowitz, N; Mistry, S; McGowan, E; Hajdu, R; Rosenbach, M; Abbadie, C; Alexander, JP; Shiao, LL; Sullivan, KM; Nargund, RP; Wyvratt, MJ; Lin, LS; DeVita, RJ Discovery of MK-4409, a Novel Oxazole FAAH Inhibitor for the Treatment of Inflammatory and Neuropathic Pain. ACS Med Chem Lett5:717-21 (2014) [PubMed] Article
Chobanian, HR; Guo, Y; Liu, P; Chioda, MD; Fung, S; Lanza, TJ; Chang, L; Bakshi, RK; Dellureficio, JP; Hong, Q; McLaughlin, M; Belyk, KM; Krska, SW; Makarewicz, AK; Martel, EJ; Leone, JF; Frey, L; Karanam, B; Madeira, M; Alvaro, R; Shuman, J; Salituro, G; Terebetski, JL; Jochnowitz, N; Mistry, S; McGowan, E; Hajdu, R; Rosenbach, M; Abbadie, C; Alexander, JP; Shiao, LL; Sullivan, KM; Nargund, RP; Wyvratt, MJ; Lin, LS; DeVita, RJ Discovery of MK-4409, a Novel Oxazole FAAH Inhibitor for the Treatment of Inflammatory and Neuropathic Pain. ACS Med Chem Lett5:717-21 (2014) [PubMed] Article