| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50028142 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1434723 (CHEMBL3382838) |
|---|
| IC50 | >33000±n/a nM |
|---|
| Citation |  Gosmini, R; Nguyen, VL; Toum, J; Simon, C; Brusq, JM; Krysa, G; Mirguet, O; Riou-Eymard, AM; Boursier, EV; Trottet, L; Bamborough, P; Clark, H; Chung, CW; Cutler, L; Demont, EH; Kaur, R; Lewis, AJ; Schilling, MB; Soden, PE; Taylor, S; Walker, AL; Walker, MD; Prinjha, RK; Nicod�me, E The discovery of I-BET726 (GSK1324726A), a potent tetrahydroquinoline ApoA1 up-regulator and selective BET bromodomain inhibitor. J Med Chem57:8111-31 (2014) [PubMed] Article Gosmini, R; Nguyen, VL; Toum, J; Simon, C; Brusq, JM; Krysa, G; Mirguet, O; Riou-Eymard, AM; Boursier, EV; Trottet, L; Bamborough, P; Clark, H; Chung, CW; Cutler, L; Demont, EH; Kaur, R; Lewis, AJ; Schilling, MB; Soden, PE; Taylor, S; Walker, AL; Walker, MD; Prinjha, RK; Nicod�me, E The discovery of I-BET726 (GSK1324726A), a potent tetrahydroquinoline ApoA1 up-regulator and selective BET bromodomain inhibitor. J Med Chem57:8111-31 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50028142 |
|---|
| n/a |
|---|
| Name | BDBM50028142 |
|---|
| Synonyms: | CHEMBL2177300 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H23ClN2O3 |
|---|
| Mol. Mass. | 434.915 |
|---|
| SMILES | C[C@H]1C[C@@H](Nc2ccc(Cl)cc2)c2cc(ccc2N1C(C)=O)-c1ccc(cc1)C(O)=O |r| |
|---|
| Structure | 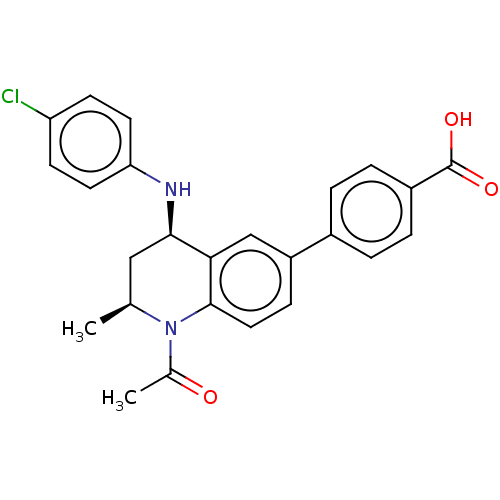
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gosmini, R; Nguyen, VL; Toum, J; Simon, C; Brusq, JM; Krysa, G; Mirguet, O; Riou-Eymard, AM; Boursier, EV; Trottet, L; Bamborough, P; Clark, H; Chung, CW; Cutler, L; Demont, EH; Kaur, R; Lewis, AJ; Schilling, MB; Soden, PE; Taylor, S; Walker, AL; Walker, MD; Prinjha, RK; Nicod�me, E The discovery of I-BET726 (GSK1324726A), a potent tetrahydroquinoline ApoA1 up-regulator and selective BET bromodomain inhibitor. J Med Chem57:8111-31 (2014) [PubMed] Article
Gosmini, R; Nguyen, VL; Toum, J; Simon, C; Brusq, JM; Krysa, G; Mirguet, O; Riou-Eymard, AM; Boursier, EV; Trottet, L; Bamborough, P; Clark, H; Chung, CW; Cutler, L; Demont, EH; Kaur, R; Lewis, AJ; Schilling, MB; Soden, PE; Taylor, S; Walker, AL; Walker, MD; Prinjha, RK; Nicod�me, E The discovery of I-BET726 (GSK1324726A), a potent tetrahydroquinoline ApoA1 up-regulator and selective BET bromodomain inhibitor. J Med Chem57:8111-31 (2014) [PubMed] Article