

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cannabinoid receptor 1 | ||
| Ligand | BDBM50258652 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1438383 (CHEMBL3383058) | ||
| Ki | 200±n/a nM | ||
| Citation |  Lucchesi, V; Hurst, DP; Shore, DM; Bertini, S; Ehrmann, BM; Allar�, M; Lawrence, L; Ligresti, A; Minutolo, F; Saccomanni, G; Sharir, H; Macchia, M; Di Marzo, V; Abood, ME; Reggio, PH; Manera, C CB2-selective cannabinoid receptor ligands: synthesis, pharmacological evaluation, and molecular modeling investigation of 1,8-Naphthyridin-2(1H)-one-3-carboxamides. J Med Chem57:8777-91 (2014) [PubMed] Article Lucchesi, V; Hurst, DP; Shore, DM; Bertini, S; Ehrmann, BM; Allar�, M; Lawrence, L; Ligresti, A; Minutolo, F; Saccomanni, G; Sharir, H; Macchia, M; Di Marzo, V; Abood, ME; Reggio, PH; Manera, C CB2-selective cannabinoid receptor ligands: synthesis, pharmacological evaluation, and molecular modeling investigation of 1,8-Naphthyridin-2(1H)-one-3-carboxamides. J Med Chem57:8777-91 (2014) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cannabinoid receptor 1 | |||
| Name: | Cannabinoid receptor 1 | ||
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 52868.96 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P21554 | ||
| Residue: | 472 | ||
| Sequence: |
| ||
| BDBM50258652 | |||
| n/a | |||
| Name | BDBM50258652 | ||
| Synonyms: | CHEMBL466651 | N-(4-Methylcyclohexyl)-1-(p-fluorobenzyl)-1,8-naphthyridin-2(1H)-on-3-carboxamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C23H24FN3O2 | ||
| Mol. Mass. | 393.454 | ||
| SMILES | CC1CCC(CC1)NC(=O)c1cc2cccnc2n(Cc2ccc(F)cc2)c1=O |(3.5,6.21,;2.17,5.44,;2.17,3.9,;.84,3.13,;-.5,3.91,;-.5,5.44,;.83,6.21,;-1.83,3.14,;-3.17,3.9,;-3.17,5.44,;-4.5,3.13,;-5.85,3.91,;-7.19,3.12,;-8.52,3.88,;-9.85,3.11,;-9.85,1.57,;-8.52,.8,;-7.18,1.56,;-5.84,.8,;-5.83,-.74,;-4.49,-1.5,;-4.49,-3.04,;-3.16,-3.8,;-1.83,-3.02,;-.49,-3.79,;-1.84,-1.5,;-3.16,-.74,;-4.49,1.57,;-3.16,.81,)| | ||
| Structure | 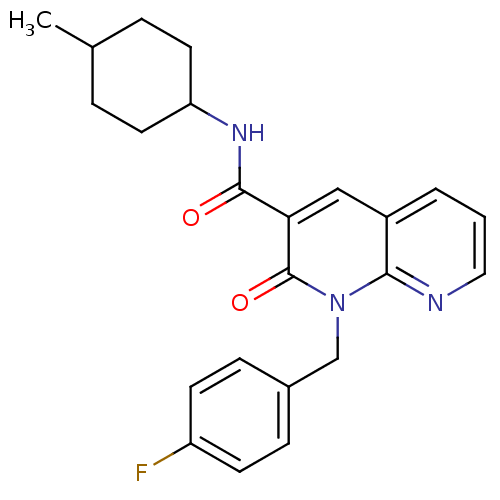
| ||