

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Coagulation factor X | ||
| Ligand | BDBM50032874 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1431778 (CHEMBL3385074) | ||
| Ki | >10000±n/a nM | ||
| Citation |  Hangeland, JJ; Friends, TJ; Rossi, KA; Smallheer, JM; Wang, C; Sun, Z; Corte, JR; Fang, T; Wong, PC; Rendina, AR; Barbera, FA; Bozarth, JM; Luettgen, JM; Watson, CA; Zhang, G; Wei, A; Ramamurthy, V; Morin, PE; Bisacchi, GS; Subramaniam, S; Arunachalam, P; Mathur, A; Seiffert, DA; Wexler, RR; Quan, ML Phenylimidazoles as potent and selective inhibitors of coagulation factor XIa with in vivo antithrombotic activity. J Med Chem57:9915-32 (2014) [PubMed] Article Hangeland, JJ; Friends, TJ; Rossi, KA; Smallheer, JM; Wang, C; Sun, Z; Corte, JR; Fang, T; Wong, PC; Rendina, AR; Barbera, FA; Bozarth, JM; Luettgen, JM; Watson, CA; Zhang, G; Wei, A; Ramamurthy, V; Morin, PE; Bisacchi, GS; Subramaniam, S; Arunachalam, P; Mathur, A; Seiffert, DA; Wexler, RR; Quan, ML Phenylimidazoles as potent and selective inhibitors of coagulation factor XIa with in vivo antithrombotic activity. J Med Chem57:9915-32 (2014) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Coagulation factor X | |||
| Name: | Coagulation factor X | ||
| Synonyms: | F10 | FA10_RABIT | ||
| Type: | PROTEIN | ||
| Mol. Mass.: | 53951.21 | ||
| Organism: | Oryctolagus cuniculus | ||
| Description: | ChEMBL_1471174 | ||
| Residue: | 490 | ||
| Sequence: |
| ||
| BDBM50032874 | |||
| n/a | |||
| Name | BDBM50032874 | ||
| Synonyms: | CHEMBL3355683 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C28H31ClF3N7O3 | ||
| Mol. Mass. | 606.039 | ||
| SMILES | OC(=O)C(F)(F)F.NC[C@H]1CC[C@@H](CC1)C(=O)N[C@@H](Cc1ccccc1)c1nc(c(Cl)[nH]1)-c1ccc2c(N)n[nH]c2c1 |r,wU:12.14,wD:9.7,18.17,(34.53,-30.97,;35.85,-31.73,;35.85,-33.26,;37.17,-30.97,;38.49,-31.73,;37.17,-29.44,;38.49,-30.19,;15.69,-45.08,;16.46,-43.74,;18,-43.74,;18.77,-45.08,;20.31,-45.08,;21.08,-43.74,;20.31,-42.42,;18.77,-42.42,;22.62,-43.74,;23.39,-45.08,;23.39,-42.42,;24.93,-42.42,;25.7,-43.74,;27.24,-43.74,;28.01,-42.42,;29.55,-42.42,;30.32,-43.74,;29.55,-45.08,;28.01,-45.08,;25.7,-41.08,;25.07,-39.67,;26.21,-38.64,;27.55,-39.42,;28.96,-38.78,;27.23,-40.92,;26.05,-37.11,;27.3,-36.21,;27.14,-34.68,;25.73,-34.05,;25.26,-32.58,;26.16,-31.33,;23.72,-32.58,;23.24,-34.05,;24.49,-34.96,;24.65,-36.49,)| | ||
| Structure | 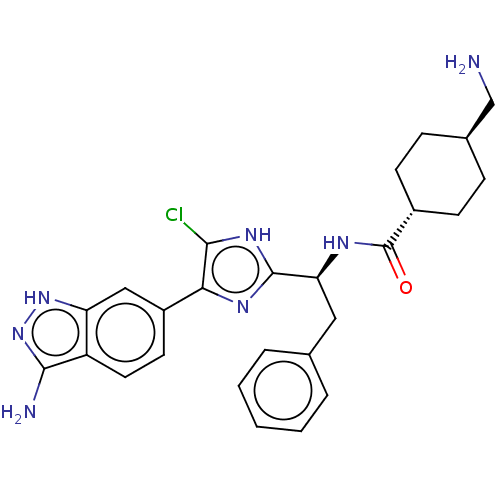
| ||