| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50052254 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1456622 (CHEMBL3370222) |
|---|
| IC50 | >5000±n/a nM |
|---|
| Citation |  Dragovich, PS; Fauber, BP; Boggs, J; Chen, J; Corson, LB; Ding, CZ; Eigenbrot, C; Ge, H; Giannetti, AM; Hunsaker, T; Labadie, S; Li, C; Liu, Y; Liu, Y; Ma, S; Malek, S; Peterson, D; Pitts, KE; Purkey, HE; Robarge, K; Salphati, L; Sideris, S; Ultsch, M; VanderPorten, E; Wang, J; Wei, B; Xu, Q; Yen, I; Yue, Q; Zhang, H; Zhang, X; Zhou, A Identification of substituted 3-hydroxy-2-mercaptocyclohex-2-enones as potent inhibitors of human lactate dehydrogenase. Bioorg Med Chem Lett24:3764-71 (2014) [PubMed] Article Dragovich, PS; Fauber, BP; Boggs, J; Chen, J; Corson, LB; Ding, CZ; Eigenbrot, C; Ge, H; Giannetti, AM; Hunsaker, T; Labadie, S; Li, C; Liu, Y; Liu, Y; Ma, S; Malek, S; Peterson, D; Pitts, KE; Purkey, HE; Robarge, K; Salphati, L; Sideris, S; Ultsch, M; VanderPorten, E; Wang, J; Wei, B; Xu, Q; Yen, I; Yue, Q; Zhang, H; Zhang, X; Zhou, A Identification of substituted 3-hydroxy-2-mercaptocyclohex-2-enones as potent inhibitors of human lactate dehydrogenase. Bioorg Med Chem Lett24:3764-71 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50052254 |
|---|
| n/a |
|---|
| Name | BDBM50052254 |
|---|
| Synonyms: | 2',6'-dichloro-4-((2-chlorophenyl)thio)-5-hydroxy-1,6-dihydro-[1,1'-biphenyl]-3(2H)-one (3) | CHEMBL3318469 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H13Cl3O2S |
|---|
| Mol. Mass. | 399.719 |
|---|
| SMILES | OC1=C(Sc2ccccc2Cl)C(=O)CC(C1)c1c(Cl)cccc1Cl |c:1| |
|---|
| Structure | 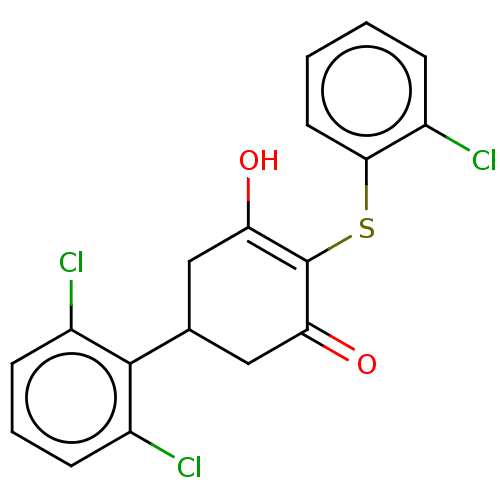
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dragovich, PS; Fauber, BP; Boggs, J; Chen, J; Corson, LB; Ding, CZ; Eigenbrot, C; Ge, H; Giannetti, AM; Hunsaker, T; Labadie, S; Li, C; Liu, Y; Liu, Y; Ma, S; Malek, S; Peterson, D; Pitts, KE; Purkey, HE; Robarge, K; Salphati, L; Sideris, S; Ultsch, M; VanderPorten, E; Wang, J; Wei, B; Xu, Q; Yen, I; Yue, Q; Zhang, H; Zhang, X; Zhou, A Identification of substituted 3-hydroxy-2-mercaptocyclohex-2-enones as potent inhibitors of human lactate dehydrogenase. Bioorg Med Chem Lett24:3764-71 (2014) [PubMed] Article
Dragovich, PS; Fauber, BP; Boggs, J; Chen, J; Corson, LB; Ding, CZ; Eigenbrot, C; Ge, H; Giannetti, AM; Hunsaker, T; Labadie, S; Li, C; Liu, Y; Liu, Y; Ma, S; Malek, S; Peterson, D; Pitts, KE; Purkey, HE; Robarge, K; Salphati, L; Sideris, S; Ultsch, M; VanderPorten, E; Wang, J; Wei, B; Xu, Q; Yen, I; Yue, Q; Zhang, H; Zhang, X; Zhou, A Identification of substituted 3-hydroxy-2-mercaptocyclohex-2-enones as potent inhibitors of human lactate dehydrogenase. Bioorg Med Chem Lett24:3764-71 (2014) [PubMed] Article