| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2Y purinoceptor 12 |
|---|
| Ligand | BDBM50100269 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1454020 (CHEMBL3367079) |
|---|
| IC50 | 4600±n/a nM |
|---|
| Citation |  Boldron, C; Besse, A; Bordes, MF; Tissandi�, S; Yvon, X; Gau, B; Badorc, A; Rousseaux, T; Barr�, G; Meneyrol, J; Zech, G; Nazare, M; Fossey, V; Pflieger, AM; Bonnet-Lignon, S; Millet, L; Briot, C; Dol, F; H�rault, JP; Savi, P; Lassalle, G; Delesque, N; Herbert, JM; Bono, F N-[6-(4-butanoyl-5-methyl-1H-pyrazol-1-yl)pyridazin-3-yl]-5-chloro-1-[2-(4-methylpiperazin-1-yl)-2-oxoethyl]-1H-indole-3-carboxamide (SAR216471), a novel intravenous and oral, reversible, and directly acting P2Y12 antagonist. J Med Chem57:7293-316 (2014) [PubMed] Article Boldron, C; Besse, A; Bordes, MF; Tissandi�, S; Yvon, X; Gau, B; Badorc, A; Rousseaux, T; Barr�, G; Meneyrol, J; Zech, G; Nazare, M; Fossey, V; Pflieger, AM; Bonnet-Lignon, S; Millet, L; Briot, C; Dol, F; H�rault, JP; Savi, P; Lassalle, G; Delesque, N; Herbert, JM; Bono, F N-[6-(4-butanoyl-5-methyl-1H-pyrazol-1-yl)pyridazin-3-yl]-5-chloro-1-[2-(4-methylpiperazin-1-yl)-2-oxoethyl]-1H-indole-3-carboxamide (SAR216471), a novel intravenous and oral, reversible, and directly acting P2Y12 antagonist. J Med Chem57:7293-316 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2Y purinoceptor 12 |
|---|
| Name: | P2Y purinoceptor 12 |
|---|
| Synonyms: | P2Y purinoceptor 12 | P2Y12_RAT | P2ry12 | P2y12 | Purinergic receptor P2Y12 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 39068.50 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1454020 |
|---|
| Residue: | 343 |
|---|
| Sequence: | MEVPGANATSANTTSIPGTSTLCSRDYKITQVLFPLLYTVLFFAGLITNSLAMRIFFQIR
SKSNFIIFLKNTVISDLLMILTFPFKILSDAKLGAGHLRTLVCQVTSVTFYFTMYISISF
LGLITIDRYLKTTRPFKTSSPSNLLGAKILSVAIWAFMFLLSLPNMILTNRRPKDKDITK
CSFLKSEFGLVWHEIVNYICQVIFWINFLIVIVCYSLITKELYRSYVRTRGSAKAPKKRV
NIKVFIIIAVFFICFVPFHFARIPYTLSQTRAVFDCNAENTLFYVKESTLWLTSLNACLD
PFIYFFLCKSFRNSLMSMLRCSTSGANKKKGQEGGDPSEETPM
|
|
|
|---|
| BDBM50100269 |
|---|
| n/a |
|---|
| Name | BDBM50100269 |
|---|
| Synonyms: | CHEMBL3325627 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H28N4O4 |
|---|
| Mol. Mass. | 472.5356 |
|---|
| SMILES | CCCC(=O)c1cnn(c1C)-c1ccc(NC(=O)c2cn(CC(O)=O)c3cc(C)c(C)cc23)cc1 |
|---|
| Structure | 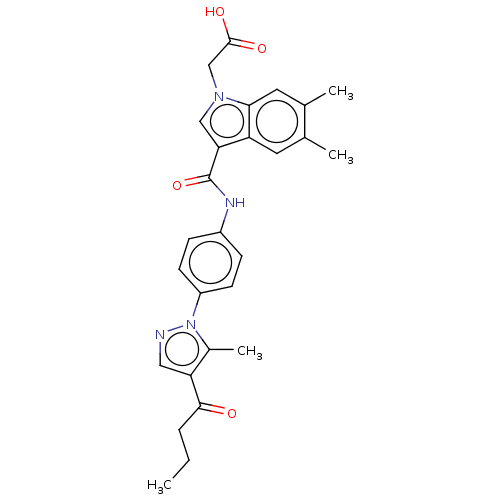
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Boldron, C; Besse, A; Bordes, MF; Tissandi�, S; Yvon, X; Gau, B; Badorc, A; Rousseaux, T; Barr�, G; Meneyrol, J; Zech, G; Nazare, M; Fossey, V; Pflieger, AM; Bonnet-Lignon, S; Millet, L; Briot, C; Dol, F; H�rault, JP; Savi, P; Lassalle, G; Delesque, N; Herbert, JM; Bono, F N-[6-(4-butanoyl-5-methyl-1H-pyrazol-1-yl)pyridazin-3-yl]-5-chloro-1-[2-(4-methylpiperazin-1-yl)-2-oxoethyl]-1H-indole-3-carboxamide (SAR216471), a novel intravenous and oral, reversible, and directly acting P2Y12 antagonist. J Med Chem57:7293-316 (2014) [PubMed] Article
Boldron, C; Besse, A; Bordes, MF; Tissandi�, S; Yvon, X; Gau, B; Badorc, A; Rousseaux, T; Barr�, G; Meneyrol, J; Zech, G; Nazare, M; Fossey, V; Pflieger, AM; Bonnet-Lignon, S; Millet, L; Briot, C; Dol, F; H�rault, JP; Savi, P; Lassalle, G; Delesque, N; Herbert, JM; Bono, F N-[6-(4-butanoyl-5-methyl-1H-pyrazol-1-yl)pyridazin-3-yl]-5-chloro-1-[2-(4-methylpiperazin-1-yl)-2-oxoethyl]-1H-indole-3-carboxamide (SAR216471), a novel intravenous and oral, reversible, and directly acting P2Y12 antagonist. J Med Chem57:7293-316 (2014) [PubMed] Article